Table of Contents |
Measurements provide much of the information that describes the behavior of matter and energy in both the macroscopic and microscopic domains of chemistry. The macroscopic domain of chemistry is the effects of chemistry on our world that is visible to our eyes. Examples would be seeing water boil or salt dissolve in water. The microscopic domain of chemistry is what is happening at the molecular or atomic level that is causing the effects of chemistry we see on the macroscopic level.
Every measurement provides three kinds of information: the size or magnitude of the measurement (a number), a standard of comparison for the measurement (a unit), and an indication of the uncertainty of the measurement. While the number and unit are explicitly represented when a quantity is written, the uncertainty is an aspect of the measurement result that is more implicitly represented and will be discussed later.
The number in the measurement can be represented in different ways, including decimal form and scientific notation. While you are familiar with decimals, you may not be familiar with scientific notation. Scientific notation is a format for writing very large or very small numbers in a shorthand manner. 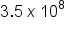 or
or 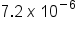 are examples of scientific notation. The “
are examples of scientific notation. The “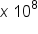 ” refers to how many spaces the decimal must be moved to convert the number in scientific notation to decimal. If the exponent is positive, the decimal is moved to the right, adding zeroes as needed. If the exponent is negative, the decimal is moved to the left, adding zeroes as needed.
” refers to how many spaces the decimal must be moved to convert the number in scientific notation to decimal. If the exponent is positive, the decimal is moved to the right, adding zeroes as needed. If the exponent is negative, the decimal is moved to the left, adding zeroes as needed.
When converting from decimal to scientific notation, you move the decimal till it is behind the first non-zero number. The number of spaces you moved will be the exponent. If you move to the right (numbers smaller than one), you will have a negative exponent. If you move to the left (numbers larger than one), you will have a positive exponent.
EXAMPLE
Convert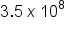 and
and 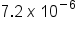 to decimal form.
to decimal form.
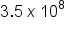 has a positive exponent, so you move the decimal to the right 8 spaces. This converts to 350,000,000 (move one spot to the right to get behind the 5 and then add 7 zeroes).
has a positive exponent, so you move the decimal to the right 8 spaces. This converts to 350,000,000 (move one spot to the right to get behind the 5 and then add 7 zeroes).
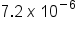 has a negative exponent, so you move the decimal to the left 6 spaces. This converts to 0.0000072 (move one spot to the left to get in front of 7 and then add 5 zeroes with a 0. In front of the five zeroes).
has a negative exponent, so you move the decimal to the left 6 spaces. This converts to 0.0000072 (move one spot to the left to get in front of 7 and then add 5 zeroes with a 0. In front of the five zeroes).
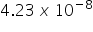 .
.
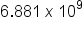 .
.
You should be able to perform calculations with scientific notation or put answers from calculations into scientific notation. When doing calculations with scientific notation, you should use your calculator. Each calculator is slightly different so make sure you know how to use your calculator to perform calculations with scientific notation.
If you don’t have a calculator or you are asked to do the calculations without a calculator, you can do scientific notation calculations without a calculator. For multiplication, you multiply the coefficients and add the exponents. For division, you divide the coefficients and subtract the exponents. After performing the calculation, convert the answer back into proper scientific notation (#.# x 10 ).
).
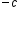 x b x 10
x b x 10 = (a x b) x 10
= (a x b) x 10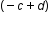
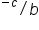 x 10
x 10 = (a/b) x 10
= (a/b) x 10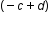
Units, such as liters, pounds, and centimeters, are standards of comparison for measurements. Without units, a number can be meaningless, confusing, or possibly life-threatening. Suppose a doctor prescribes phenobarbital to control a patient’s seizures and states a dosage of “100” without specifying units. Not only will this be confusing to the medical professional giving the dose, but the consequences can be dire: 100 mg given three times per day can be effective as an anticonvulsant, but a single dose of 100 g is more than 10 times the lethal amount. The measurement units for seven fundamental properties, SI Units, are listed below.
| Property Measured | Name of Unit | Symbol of Unit |
|---|---|---|
| length | meter | m |
| mass | kilogram | kg |
| time | second | s |
| temperature | kelvin | K |
| electric current | ampere | A |
| amount of substance | mole | mol |
| luminous intensity | candela | cd |
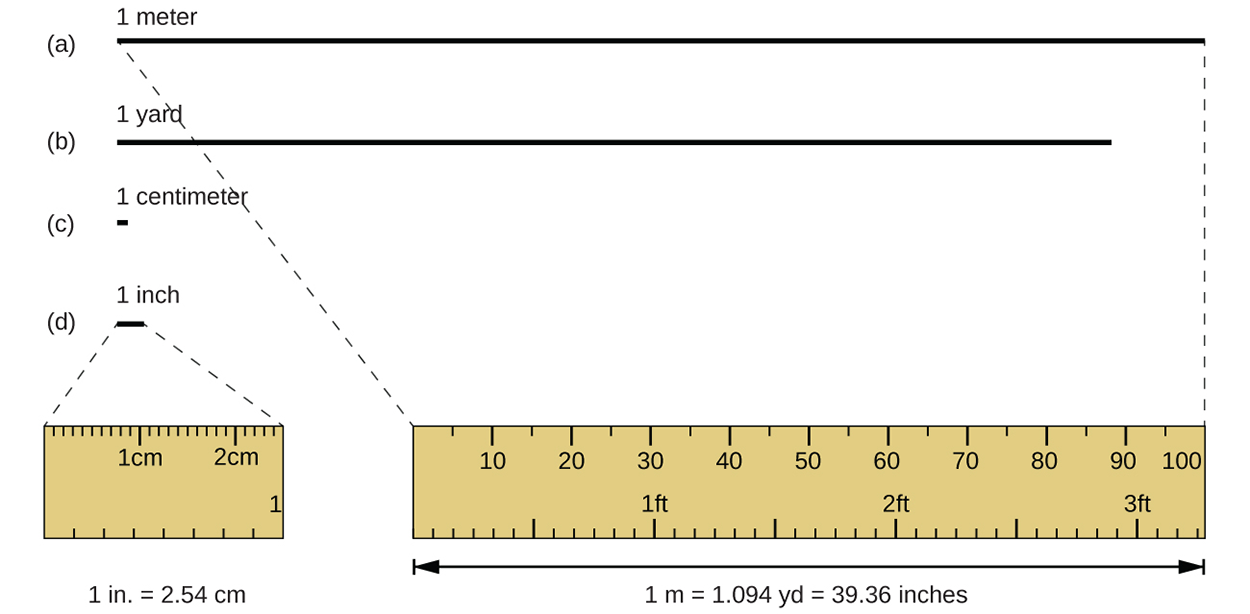
The standard unit of length is the meter (m). A meter is equal to 1.094 yards or 39.36 inches. An inch is equal to 2.54 cm. The standard unit of mass in the SI system is the kilogram (kg). One kilogram is equal to 2.2046 pounds. The SI unit of temperature is the kelvin (K) and is written without a degree symbol (°). The SI base unit of time is the second (s).
We can derive many other units from the seven SI base units. For example, we can use the base unit of length to define a unit of volume, and the base units of mass and length to define a unit of density.
Volume is the measure of the amount of space occupied by an object. The standard SI unit of volume is defined by the base unit of length. The standard volume is a cubic meter (m3), a cube with an edge length of exactly one meter. A liter (L) is the more common name for the cubic decimeter. One liter is about 1.06 quarts. A cubic centimeter (cm3) is the abbreviation cc (for cubic centimeter) and is often used by health professionals. A cubic centimeter is equivalent to a milliliter (mL) and is 1/1000 of a liter.
The density of a substance is the ratio of the mass of a sample of the substance to its volume and the units of density are defined by the base units of mass and length. The SI unit for density is the kilogram per cubic meter (kg/m3). However, we usually use grams per cubic centimeter (g/cm3) for the densities of solids and liquids, and grams per liter (g/L) for gases.
The simplest method to determine the density is to find the mass and volume of the object and then divide the mass of the sample by its volume.
EXAMPLE
A brick of gold weighs 12,400 grams and has a volume of 643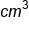 . What is the density of the gold?
. What is the density of the gold?
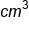 = 19.3 g/
= 19.3 g/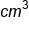
Source: THIS TUTORIAL HAS BEEN ADAPTED FROM OPENSTAX “CHEMISTRY: ATOMS FIRST 2E”. ACCESS FOR FREE AT Chemistry: Atoms First 2e. LICENSE: CREATIVE COMMONS ATTRIBUTION 4.0 INTERNATIONAL