Table of Contents |
As mentioned in a previous lesson in this unit, our atmosphere is huge. It extends over 10,000 km above the surface of the Earth. The lowest layer of our atmosphere is the troposphere, which extends from the surface of the Earth to about 12 km above the surface of the Earth (about 3 km over the height of Everest). While some contaminants do get into the upper levels of the atmosphere (Freon which destroys the ozone layer in the stratosphere), most of the contaminants that cause what we call pollution are found right above the surface of the Earth.
Recall that air pollutants are generally classified as either primary air pollutants or secondary air pollutants. Primary air pollutants are released directly into the atmosphere by humans or nature.
EXAMPLE
These include sulfur dioxide (SO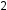 ) and carbon dioxide (CO
) and carbon dioxide (CO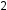 ), which are released from the burning of fossil fuels and volcanic eruptions.
), which are released from the burning of fossil fuels and volcanic eruptions.
Secondary air pollutants are pollutants that result from a chemical reaction of a primary air pollutant.
EXAMPLE
Sulfur trioxide (SO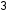 ), sulfuric acid (H
), sulfuric acid (H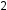 SO
SO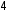 ), and ozone (O
), and ozone (O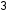 ) are examples of secondary air pollutants. Sulfur trioxide and sulfuric acid arise from the oxidation of sulfur dioxide, while ozone comes from the oxidation of oxygen (O
) are examples of secondary air pollutants. Sulfur trioxide and sulfuric acid arise from the oxidation of sulfur dioxide, while ozone comes from the oxidation of oxygen (O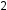 ) gas.
) gas.
Pollutants refer to any material that contaminates something (such as water or the atmosphere). Most of what we call pollutants are usually very small particulate pieces of matter or gaseous chemicals released into the atmosphere from either natural or artificial sources (like factories or cars). Particulate matter is microscopic solid or liquid pieces of matter suspended in the air. So, pollution is a suspension, where air acts as the solvent.
Air pollution can be classified as outdoor and indoor air pollution, which is just what it sounds like. Outdoor air pollution is pollutants released outdoors, and indoor air pollution is pollutants released inside of homes and buildings. Let's look at indoor air pollution first.
EXAMPLE
Indoor air pollution can be as simple as dust, cigarette (or other tobacco) smoke, mold, or mildew. Or it can be more complex such as allergens, volatile organic compounds, carbon monoxide, and radon gas.So, where do all these pollutants come from? Sources of indoor pollution are much easier to identify. Tobacco smoke obviously comes from people smoking indoors. Mold and mildew come from sources of water in walls or attic, plus bathrooms, kitchens, laundry rooms that are filled with moisture. Dust and skin cells come from humans and human activity. Pet dander comes from pets. Some allergens can come from pets, humans (perfumes, colognes, soaps, detergents, etc.), and even pests, such as rodents and roaches.
Volatile organic compounds can come from freshly painted surfaces, new furniture, and even clothing treated with chemicals (such as water repellent chemicals). Building material can also release chemicals. Carbon monoxide is often the result of any open flame or heating source, such as grills, hot water heaters, space heaters, portable generators, dryers, stoves, and even fireplaces.
IN CONTEXT
Radon is a very deadly indoor pollutant. While radon can come from building materials, some water supplies, and even natural gas, the most common source of radon radiation is a slow seepage from the ground into the house through tiny cracks or breaks in the foundation. This is much more common in houses with basements, where the ground is dug out for the basement and radon can be released by breaking the rocks. Additionally, the sides of the basement offer an entry into the basement in addition to the floor-only route for houses without a basement.
Outdoor air pollution can come from a variety of sources. Major sources of pollution include manufacturing facilities that release potential pollutants into the air through smokestacks or discharged into water supplies. Power plants are another major source of pollutants. All of these manufacturing, industrial plants, and power plants have filtration units that should filter out these chemicals before they are released into the atmosphere, but some get through. Some are “benign,” but can then form secondary pollutants via chemical reactions. Nitrogen oxides, carbon dioxide, and sulfur oxides are the most common of these pollutants.
Cars, trains, buses, and planes are a major source of outdoor air pollutants. Even modern cars with catalytic converters that convert more pollutants like hydrocarbons and carbon monoxide into less polluting carbon dioxide and water, still contribute to outdoor air pollution. Additionally, carbon dioxide is a greenhouse gas that contributes to climate change.
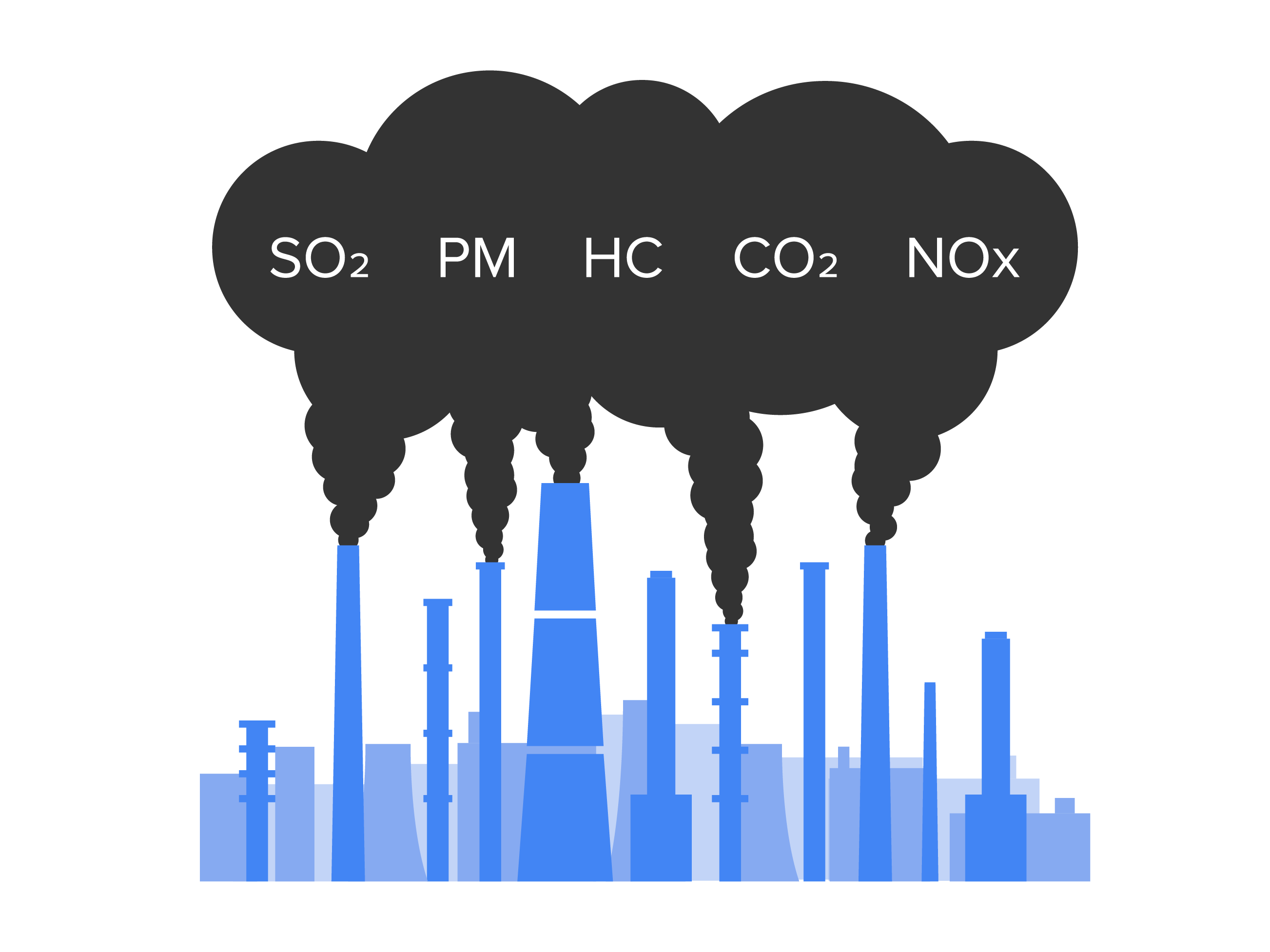
Major construction can release multiple pollutants such as volatile organic compounds, radon gas, carbon dioxide, fine dust, and other particulate matter that contribute to pollution. Domesticated animals, such as cattle, sheep, and horses, can contribute ammonia to the atmosphere from their manure. Fertilizers and pesticides can also release contaminants into the atmosphere. Waste and landfills contribute methane, a greenhouse gas, into the atmosphere.
There are many natural sources of pollutants including radon released from rocks in the soil. Forest fires, which are becoming more and more prevalent, can release a large amount of small particulate matter that can be cancer-causing. Lightning can cause the oxidation of nitrogen into nitric oxide (NO) and oxygen into ozone (O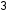 ). Volcanoes can emit incredible amounts of sulfur dioxide (SO
). Volcanoes can emit incredible amounts of sulfur dioxide (SO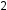 ) into the atmosphere, like Mount Pinatubo when it erupted in 1991. That eruption released an estimated 20 million tons of sulfur dioxide in a single day.
) into the atmosphere, like Mount Pinatubo when it erupted in 1991. That eruption released an estimated 20 million tons of sulfur dioxide in a single day.
The most common air pollutants are oxides of nitrogen and sulfur, carbon monoxide, radon, ozone in the troposphere, and greenhouse gases. We will talk about stratospheric ozone and greenhouse gases in the next two lessons in this unit. For this section, we will focus on the sulfur and nitrogen oxides, carbon monoxide, radon, and ozone near the surface of the Earth.
Sulfur oxides, especially sulfur dioxide (SO2), is a huge contaminant. About ¾ of this pollutant comes from power plants with the rest mostly coming from industrial facilities. Sulfur dioxide will react with water in our atmosphere to form sulfurous acid, which is the main source of acid rain. Acid rain can cause damage to structures and statues by reacting with the marble (calcium carbonate, CaCO3) to form soluble materials, which are washed away, deteriorating the statue.
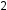 (g) + H
(g) + H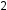 O(g) ⟶ H
O(g) ⟶ H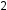 SO
SO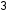 (aq)
(aq)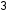 (s) + H
(s) + H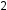 SO3(aq) ⟶ CaSO3(aq) + CO
SO3(aq) ⟶ CaSO3(aq) + CO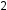 (g) + H
(g) + H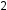 O(l)
O(l)
The image above illustrates that a statue made from carbonate compounds such as limestone and marble typically weather slowly over time due to the actions of water, thermal expansion, and contraction. However, pollutants like sulfur dioxide can accelerate weathering. As the concentration of air pollutants increases, deterioration of limestone (marble) occurs more rapidly.
Nitrogen oxides (NO and NO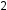 ) are often referred to as NO
) are often referred to as NO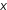 pollutants (for NO
pollutants (for NO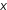 , where x is the number of nitrogens). Nitrogen dioxide (NO
, where x is the number of nitrogens). Nitrogen dioxide (NO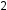 ) is the most common of these NO
) is the most common of these NO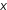 pollutants. Nitrogen (N
pollutants. Nitrogen (N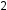 ) in the air can be converted to NO
) in the air can be converted to NO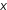 by oxidation reactions in the atmosphere, which can be catalyzed by exhaust from airplanes or lightning. NO
by oxidation reactions in the atmosphere, which can be catalyzed by exhaust from airplanes or lightning. NO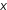 pollutants can react with volatile organic compounds to form ozone, which contributes to smog. They can also form nitrous and nitric acid and nitrogen dioxide can cause respiratory problems in humans.
pollutants can react with volatile organic compounds to form ozone, which contributes to smog. They can also form nitrous and nitric acid and nitrogen dioxide can cause respiratory problems in humans.
EXAMPLE
The main sources of NO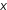 pollutants are from vehicles that run on fossil fuels (boats, cars, trucks, off-road vehicles, and construction equipment), power plants, and industrial plants. Other minor sources include any open flame, agriculture, and other miscellaneous emissions.
pollutants are from vehicles that run on fossil fuels (boats, cars, trucks, off-road vehicles, and construction equipment), power plants, and industrial plants. Other minor sources include any open flame, agriculture, and other miscellaneous emissions.
Carbon monoxide is a pollutant that comes mainly from vehicle exhaust. The advent of the catalytic converter, such as the one in the below image, has cut down greatly on the amount of carbon monoxide released into the atmosphere, but even with catalytic converters, vehicles still release carbon monoxide into the atmosphere.
IN CONTEXT
Any open flame or burning of natural gas can also release carbon monoxide. Carbon monoxide detectors in the home are a very important defense against accidental carbon monoxide poisoning. Carbon monoxide can cause respiratory issues and even lead to death at higher concentrations.

Radon gas is a radioactive gas and accounts for over 50% of the radiation exposure that humans experience each year.
As uranium undergoes alpha decay, it will convert from uranium (atomic number 92) to thorium (atomic number 90) to radium (atomic number 88) to radon (atomic number 86) to polonium (atomic number 84), and finally to lead (atomic number 82). Lead is not radioactive so does not decay any further normally. Of all these, only radon is a gas.
Uranium is a naturally occurring element found in the rocks and solid matter of the Earth. Uranium is actually 40 times more common than silver and 500 times more common than gold. Uranium is found in sedimentary rocks and in granite. As the uranium breaks down, it will eventually form radon. Since radon is a gas, when it forms, it will escape from the rock (if there are cracks for it to escape) and get into our water supply, our homes, or the atmosphere.
As mentioned above, NO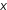 pollutants can react with volatile organic compounds to form ozone, which contributes to smog. This reaction is usually catalyzed by sunlight. Ozone is very toxic to humans and can cause breathing problems. Ozone near the surface of the Earth can also be damaging to plants and animals. Ozone is much more toxic than arsenic or cyanide.
pollutants can react with volatile organic compounds to form ozone, which contributes to smog. This reaction is usually catalyzed by sunlight. Ozone is very toxic to humans and can cause breathing problems. Ozone near the surface of the Earth can also be damaging to plants and animals. Ozone is much more toxic than arsenic or cyanide.
A typical pattern is for nitric oxide (NO) and hydrocarbons (HC) to be emitted by cars. The nitric oxide is oxidized to nitrogen dioxide by sunlight. The nitrogen dioxide then reacts with volatile organic compounds to form ozone. The ozone then creates a haze or smog. Smog used to be a product of burning coal, which created a smoke fog, which we called smog. But today most smog is a result of ozone near to the surface of the Earth.
In the pattern described above, NO and HC build up in the morning. During the later morning to early afternoon, they are converted to NO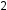 , which then reacts with volatile organic compounds in the presence of sunlight in the mid-afternoon to create that haze or smog. Tall buildings, urban sprawl, and large amounts of traffic in big cities provide for a perfect environment to promote the formation of smog, especially if the city sits in a valley or is surrounded by hills, which traps the smog.
, which then reacts with volatile organic compounds in the presence of sunlight in the mid-afternoon to create that haze or smog. Tall buildings, urban sprawl, and large amounts of traffic in big cities provide for a perfect environment to promote the formation of smog, especially if the city sits in a valley or is surrounded by hills, which traps the smog.
Source: THIS TUTORIAL HAS BEEN ADAPTED FROM OPENSTAX “CHEMISTRY: ATOMS FIRST 2E”. ACCESS FOR FREE AT Chemistry: Atoms First 2e. LICENSE: CREATIVE COMMONS ATTRIBUTION 4.0 INTERNATIONAL