Table of Contents |
Recall that plasma membranes are selectively permeable—they allow some substances to pass through, but not others. If they were to lose this selectivity, the cell would no longer be able to sustain itself, and it would be destroyed. Some cells require larger amounts of specific substances. They must have a way of obtaining these materials from extracellular fluids. This may happen passively as certain materials move back and forth, or the cell may have special mechanisms that facilitate transport.
Some materials are so important to a cell that it spends some of its energy hydrolyzing adenosine triphosphate (ATP) to obtain these materials. Red blood cells use some of their energy doing just that. Most cells spend the majority of their energy maintaining an imbalance of sodium and potassium ions between the cell's interior and exterior, as well as on protein synthesis.
The most direct forms of membrane transport are passive. Passive transport is a naturally occurring phenomenon and does not require the cell to exert any of its energy to accomplish the movement. In passive transport, substances move from an area of higher concentration to an area of lower concentration. A physical space in which there is a single substance concentration range has a concentration gradient.
Diffusion is a passive process of transport. A single substance moves from a high concentration area to a low concentration area until the concentration is equal across a space.
You are familiar with diffusion of substances through the air. For example, think about someone opening a bottle of perfume in a room filled with people. The perfume is at its highest concentration in the bottle. Its lowest concentration is at the room's edges. The perfume vapor will diffuse, or spread away, from the bottle, and gradually, increasingly more people will smell the perfume as it spreads.
Materials move within the cell’s cytosol by diffusion, and certain materials move through the plasma membrane by diffusion. Diffusion expends no energy. On the contrary, concentration gradients are a form of potential energy that dissipates as the gradient is eliminated.
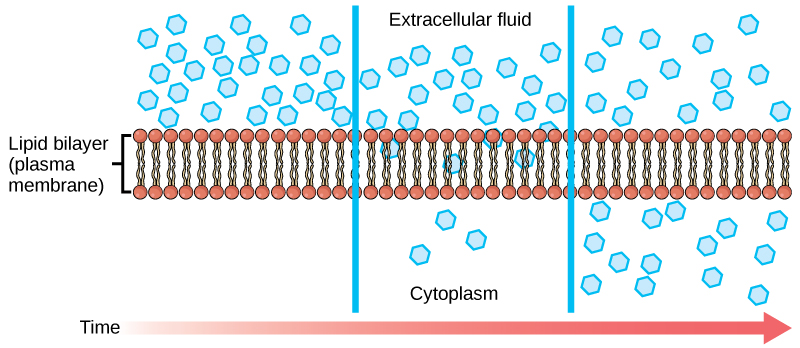
Each separate substance in a medium, such as the extracellular fluid, has its own concentration gradient, independent of other materials' concentration gradients. In addition, each substance will diffuse according to that gradient. Within a system, there will be different diffusion rates of various substances in the medium.
Molecules move constantly in a random manner at a rate that depends on their mass, their environment, and the amount of thermal energy they possess, which in turn is a function of temperature. This movement accounts for molecule diffusion through whatever medium in which they are localized. A substance moves into any space available to it until it evenly distributes itself throughout. After a substance has diffused completely through a space, removing its concentration gradient, molecules will still move around in the space, but there will be no net movement of the number of molecules from one area to another. We call this lack of a concentration gradient in which the substance has no net movement “dynamic equilibrium.”
Although diffusion will proceed in the presence of a substance's concentration gradient, several factors affect the diffusion rate.
EXAMPLE
An example of this is a person experiencing dehydration. As the body’s cells lose water, the diffusion rate decreases in the cytoplasm, and the cells’ functions deteriorate. Neurons tend to be very sensitive to this effect. Dehydration frequently leads to unconsciousness and possibly coma because of the decrease in diffusion rate within the cells.In facilitated transport, or facilitated diffusion, materials diffuse across the plasma membrane with the help of membrane proteins. A concentration gradient exists that would allow these materials to diffuse into the cell without expending cellular energy. However, these materials are polar molecule ions that the cell membrane's hydrophobic parts repel. Facilitated transport proteins shield these materials from the membrane's repulsive force, allowing them to diffuse into the cell.
The transported material first attaches to protein or glycoprotein receptors on the plasma membrane's exterior surface. This allows removal of material from the extracellular fluid that the cell needs. The substances then pass to specific integral proteins that facilitate their passage. Some of these integral proteins form a pore or channel through the phospholipid bilayer. Others are carrier proteins that bind with the substance and aid its diffusion through the membrane.
The integral proteins involved in facilitated transport are transport proteins and they function as either channels or carriers for the material. In both cases, they are transmembrane proteins.
Channels are specific for a transported substance. Channel proteins have hydrophilic domains exposed to the intracellular and extracellular fluids. In addition, they have a hydrophilic channel through their core that provides a hydrated opening through the membrane layers. Passage through the channel allows polar compounds to avoid the plasma membrane's nonpolar central layer that would otherwise slow or prevent their entry into the cell. Aquaporins are channel proteins that allow water to pass through the membrane at a very high rate.
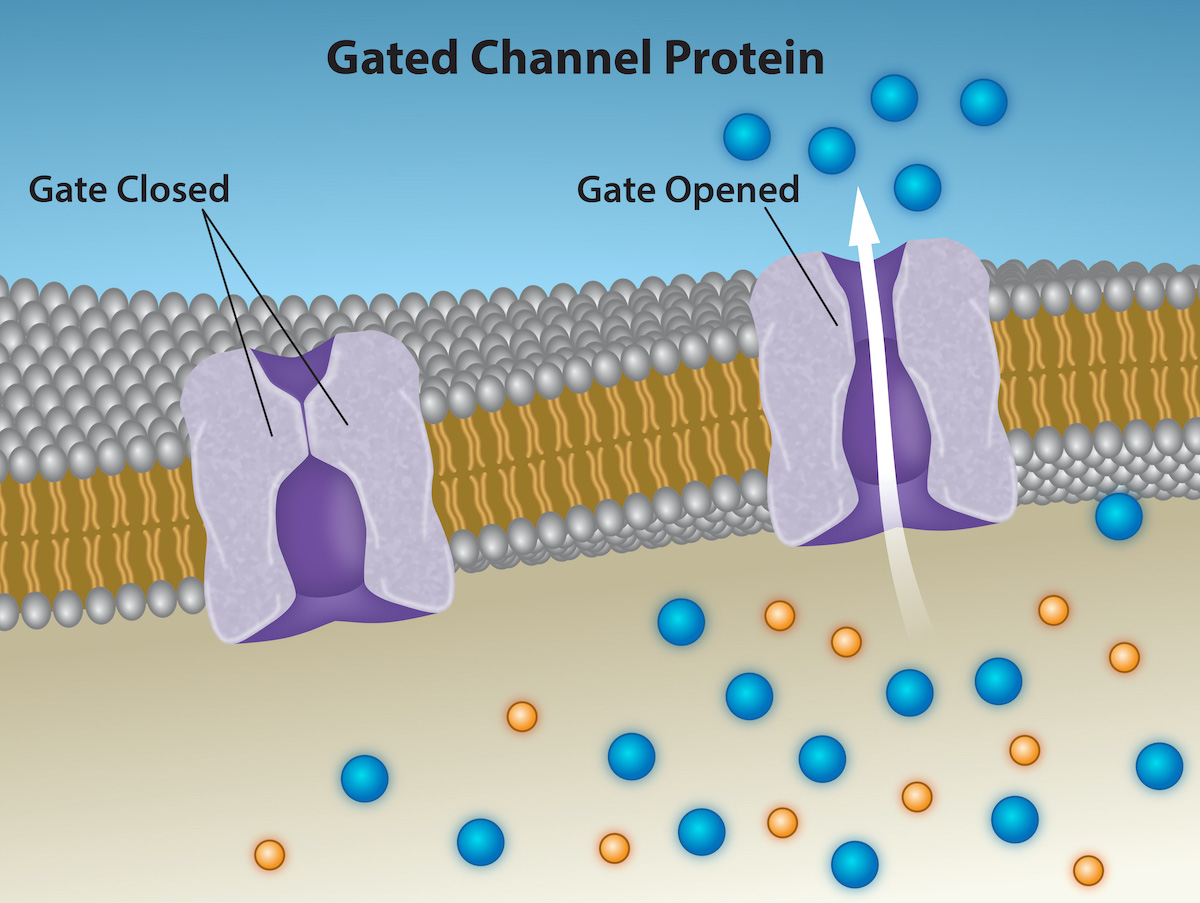
Channel proteins are either open at all times or they are “gated,” which controls the channel's opening. When a particular ion attaches to the channel protein, it may control the opening, or other mechanisms or substances may be involved. In some tissues, sodium and chloride ions pass freely through open channels; in other tissues, a gate must open to allow passage. An example of this occurs in the kidney, where there are both channel forms in different parts of the renal tubules.
Cells involved in transmitting electrical impulses, such as nerve and muscle cells, have gated channels for sodium, potassium, and calcium in their membranes. Opening and closing these channels changes the relative concentrations on opposing sides of the membrane of these ions, resulting in facilitating electrical transmission along membranes (in the case of nerve cells) or in muscle contraction (in the case of muscle cells).
Another type of protein embedded in the plasma membrane is a carrier protein. This aptly named protein binds a substance and triggers a change of its own shape (a conformational change), moving the bound molecule from the cell's outside to its interior; depending on the gradient, the material may move in the opposite direction. Carrier proteins are typically specific for a single substance. This selectivity adds to the plasma membrane's overall selectivity.
Scientists poorly understand the exact mechanism for the change of shape. Proteins can change shape when their hydrogen bonds are affected, but this may not fully explain this mechanism.
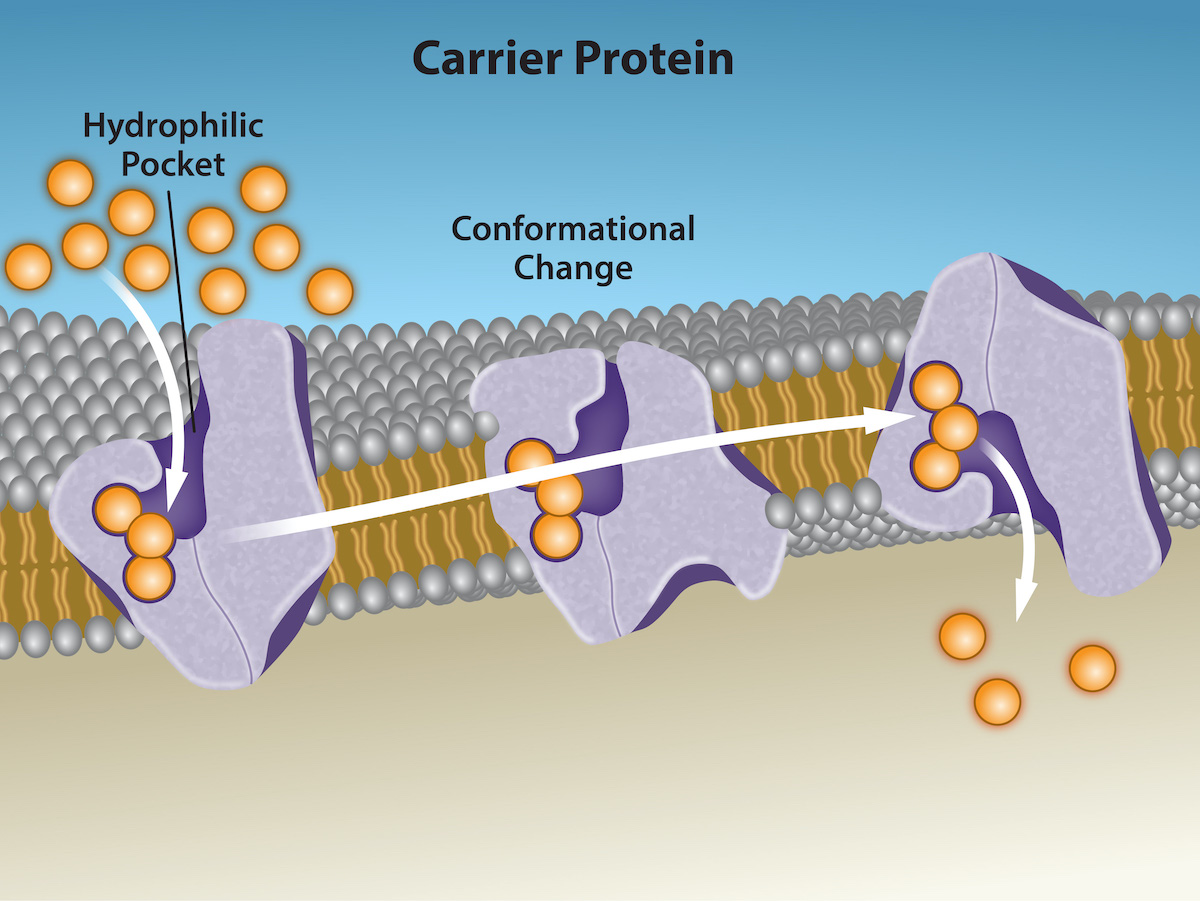
An example of this process occurs in the kidney. In one part, the kidney filters glucose, water, salts, ions, and amino acids that the body requires. This filtrate, which includes glucose, then reabsorbs in another part of the kidney. Because there are only a finite number of carrier proteins for glucose, if more glucose is present than the proteins can handle, the excess is not transported, and the body excretes this through urine. In a diabetic individual, the term is “spilling glucose into the urine.”
Osmosis is the movement of free water molecules through a semipermeable membrane according to the water's concentration gradient across the membrane. Whereas diffusion transports material across membranes and within cells, osmosis transports only water across a membrane, and the membrane limits the solutes' diffusion in the water. Not surprisingly, the aquaporins that facilitate water movement play a large role in osmosis, most prominently in red blood cells and the membranes of kidney tubules.
Water, like other substances, moves from an area of high concentration of free water molecules to one of low free water molecule concentration. An obvious question is: What makes water move at all?
Imagine a beaker with a semipermeable membrane separating the two sides or halves. On both sides of the membrane, the water level is the same, but there are different dissolved substance, or solute, concentrations that cannot cross the membrane (otherwise the solute crossing the membrane would balance concentrations on each side). If the solution's volume on both sides of the membrane is the same, but the solute's concentrations are different, then there are different amounts of water, the solvent, on either side of the membrane.
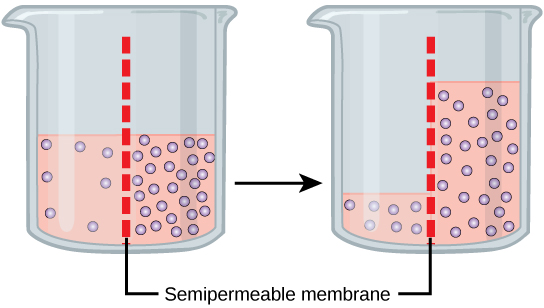
Returning to the beaker example, recall that it has a solute mixture on either side of the membrane. A principle of diffusion is that the molecules move around and will spread evenly throughout the medium if they can. However, only the material capable of getting through the membrane will diffuse through it. In this example, the solute cannot diffuse through the membrane, but the water can. Water has a concentration gradient in this system. Thus, water will diffuse down its concentration gradient, crossing the membrane to the side where it is less concentrated. This diffusion of water through the membrane—osmosis—will continue until the water's concentration gradient goes to zero or until the water's hydrostatic pressure balances the osmotic pressure. Osmosis proceeds constantly in living systems.
Tonicity describes how an extracellular solution can change a cell's volume by affecting osmosis. A solution's tonicity often directly correlates with the solution's osmolarity, which is the solution's total solute concentration. A solution with low osmolarity has a greater number of water molecules relative to the number of solute particles. A solution with high osmolarity has fewer water molecules with respect to solute particles.
In a situation in which a membrane permeable to water, though not to the solute, separates two different osmolarities, water will move from the membrane's side with lower osmolarity (and more water) to the side with higher osmolarity (and less water). This effect makes sense if you remember that the solute cannot move across the membrane, and thus the only component in the system that can move—the water—moves along its own concentration gradient.
An important distinction that concerns living systems is that osmolarity measures the number of particles (which may be molecules) in a solution. Therefore, a solution that is cloudy with cells may have a lower osmolarity than a solution that is clear, if the second solution contains more dissolved molecules than there are cells.
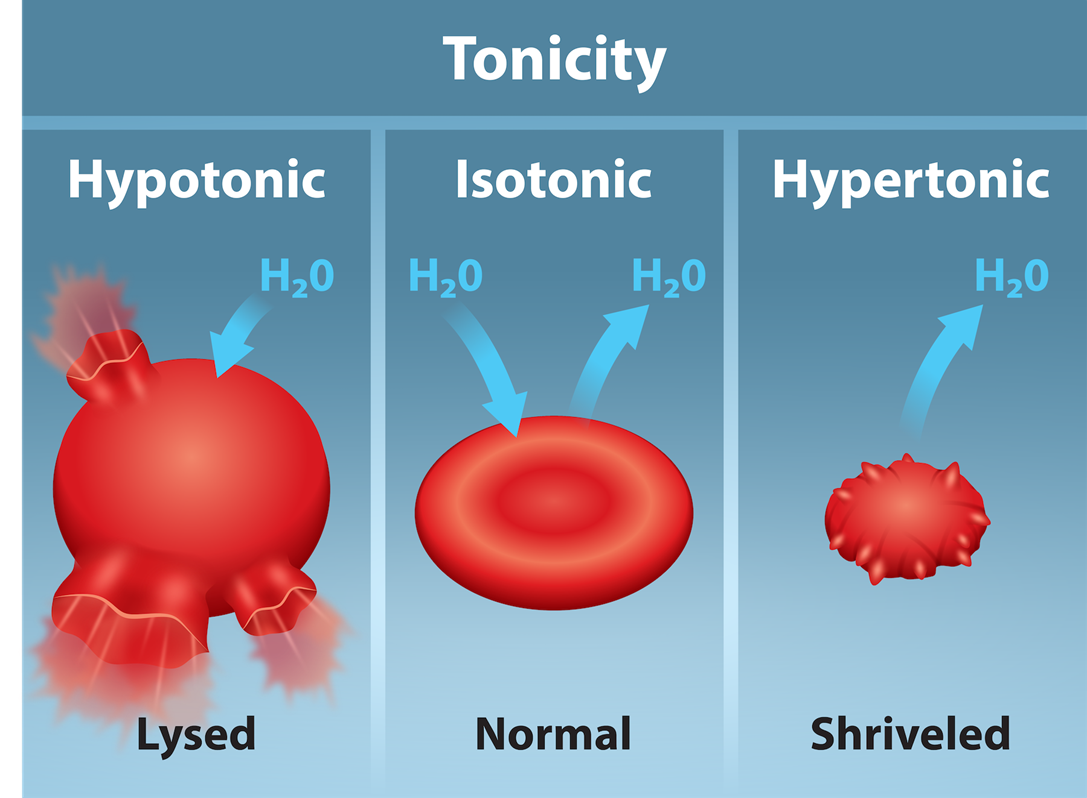
Scientists use three terms—hypotonic, isotonic, and hypertonic—to relate the cell's osmolarity to the extracellular fluid's osmolarity that contains the cells.
| Solution Type | Description |
|---|---|
| Hypotonic Solutions | In a hypotonic solution, the extracellular fluid has lower osmolarity than the fluid inside the cell, and water enters the cell. In living systems, the point of reference is always the cytoplasm, so the prefix hypo- means that the extracellular fluid has a lower solute concentration, or a lower osmolarity, than the cell cytoplasm. It also means that the extracellular fluid has a higher water concentration in the solution than does the cell. In this situation, water will follow its concentration gradient and enter the cell. |
| Hypertonic Solutions | As for a hypertonic solution, the prefix hyper- refers to the extracellular fluid having a higher osmolarity than the cell’s cytoplasm; therefore, the fluid contains less water than the cell does. Because the cell has a relatively higher water concentration, water will leave the cell. |
| Isotonic Solutions | In an isotonic solution, the extracellular fluid has the same osmolarity as the cell. If the cell's osmolarity matches that of the extracellular fluid, there will be no net movement of water into or out of the cell, although water will still move in and out. |
Blood cells in hypertonic, isotonic, and hypotonic solutions take on characteristic appearances, as shown below.
SOURCE: THIS TUTORIAL HAS BEEN ADAPTED FROM OPENSTAX “BIOLOGY 2E”. ACCESS FOR FREE AT OPENSTAX.ORG/BOOKS/BIOLOGY-2E/PAGES/1-INTRODUCTION. LICENSE: CREATIVE COMMONS ATTRIBUTION 4.0 INTERNATIONAL.