Table of Contents |
The pancreas is a long, slender organ, most of which is located posterior to the bottom half of the stomach. Although it is primarily an exocrine gland that secretes a variety of digestive enzymes, the pancreas has an endocrine function. Its pancreatic islets—clusters of cells formerly known as the islets of Langerhans—secrete the hormones glucagon, insulin, somatostatin, and pancreatic polypeptide (PP).
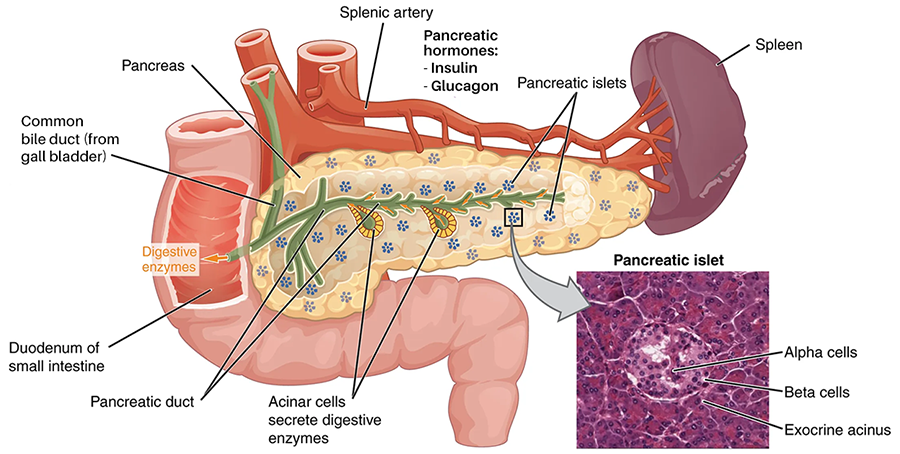
The pancreatic islets each contain four varieties of cells:
- Alpha cells produce the hormone glucagon. They make up approximately 20 percent of each islet. Glucagon plays an important role in blood glucose regulation; low blood glucose levels stimulate its release.
- Beta cells produce the hormone insulin. They make up approximately 75 percent of each islet. Elevated blood glucose levels stimulate the release of insulin.
- Delta cells account for four percent of the islet cells and secrete the peptide hormone somatostatin. Recall that somatostatin is also released by the hypothalamus (as GHIH), and the stomach and intestines also secrete it. An inhibiting hormone, pancreatic somatostatin, inhibits the release of both glucagon and insulin.
- PP cells account for about one percent of islet cells and secrete pancreatic polypeptide (PP) hormone. They are thought to play a role in appetite, as well as in the regulation of pancreatic exocrine and endocrine secretions. Pancreatic polypeptide, released following a meal, may reduce further food consumption; however, it is also released in response to fasting, and its exact role is uncertain.
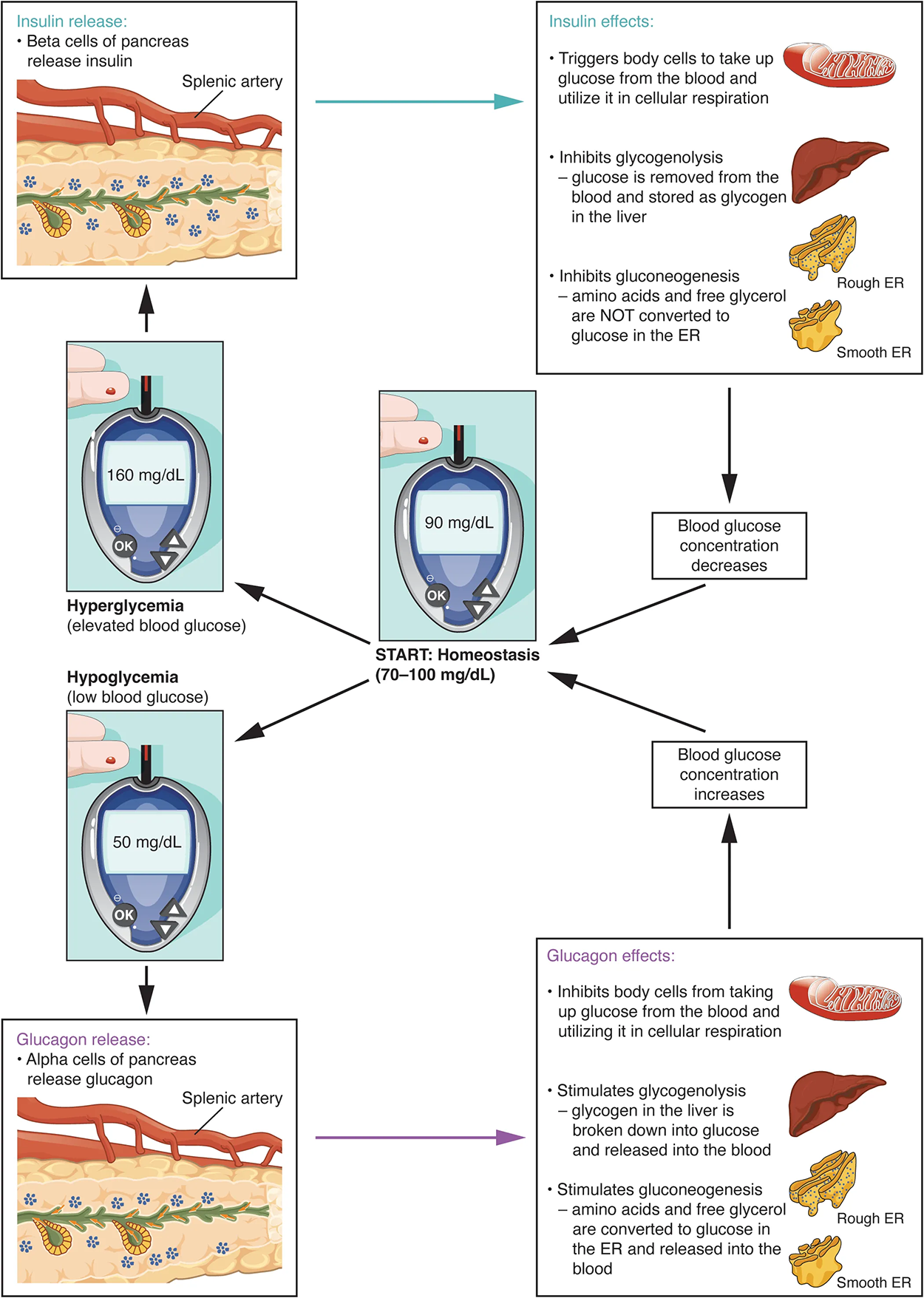
Glucose is required for cellular respiration and is the preferred fuel for all body cells. The body derives glucose from the breakdown of the carbohydrate-containing foods and drinks we consume. Glucose not immediately taken up by cells for fuel can be stored by the liver and muscles as glycogen or converted to triglycerides and stored in the adipose tissue. Hormones regulate both the storage and the utilization of glucose as required. Receptors located in the pancreas sense blood glucose levels, and subsequently, the pancreatic cells secrete glucagon or insulin to maintain normal levels.
Receptors in the pancreas can sense the decline in blood glucose levels, such as during periods of fasting or during prolonged labor or exercise. In response, the alpha cells of the pancreas secrete the hormone glucagon, which has several effects:
The figure and steps below summarize what happens as blood sugar levels change, beginning from a starting blood glucose level of 90 mg/dL that is labeled Start: homeostasis (70–100 mg/dL). From this starting point, blood sugar can increase or decrease.
Of course, blood glucose fluctuates in both directions. The step by step below describes another way that homeostasis can be maintained.
The primary function of insulin is to facilitate the uptake of glucose into body cells. Red blood cells, as well as cells of the brain, liver, kidneys, and the lining of the small intestine, do not have insulin receptors on their cell membranes and do not require insulin for glucose uptake. Although all other body cells do require insulin if they are to take glucose from the bloodstream, skeletal muscle cells and adipose cells are the primary targets of insulin.
The presence of food in the intestine triggers the release of gastrointestinal tract hormones such as glucose-dependent insulinotropic peptide (previously known as gastric inhibitory peptide). This is, in turn, the initial trigger for insulin production and secretion by the beta cells of the pancreas. Once nutrient absorption occurs, the resulting surge in blood glucose levels further stimulates insulin secretion.
Precisely how insulin facilitates glucose uptake is not entirely clear. However, insulin appears to activate a tyrosine kinase receptor, triggering the phosphorylation of many substrates within the cell. These multiple biochemical reactions converge to support the movement of intracellular vesicles containing facilitative glucose transporters to the cell membrane. In the absence of insulin, these transport proteins are normally recycled slowly between the cell membrane and the cell interior. Insulin triggers the rapid movement of a pool of glucose transporter vesicles to the cell membrane, where they fuse and expose the glucose transporters to the extracellular fluid. The transporters then move glucose by facilitated diffusion into the cell interior.
Insulin also reduces blood glucose levels by stimulating glycolysis, the metabolism of glucose for the generation of ATP. Moreover, it stimulates the liver to convert excess glucose into glycogen for storage, and it inhibits enzymes involved in glycogenolysis and gluconeogenesis. Finally, insulin promotes triglyceride and protein synthesis. The secretion of insulin is regulated through a negative feedback mechanism. As blood glucose levels decrease, further insulin release is inhibited. The pancreatic hormones are summarized in the table below.
| Associated Hormones | Chemical Class | Effect |
|---|---|---|
| Insulin (beta cells) | Protein | Reduces blood glucose levels |
| Glucagon (alpha cells) | Protein | Increases blood glucose levels |
| Somatostatin (delta cells) | Protein | Inhibits insulin and glucagon release |
| Pancreatic polypeptide (PP cells) | Protein | Role in appetite |
Recall that the hypothalamus, part of the diencephalon of the brain, sits inferior and somewhat anterior to the thalamus. Inferior but somewhat posterior to the thalamus is the pineal gland, a tiny endocrine gland whose functions are not entirely clear. The pinealocyte cells that make up the pineal gland are known to produce and secrete the amine hormone melatonin, which is derived from serotonin.
The secretion of melatonin varies according to the level of light received from the environment. When photons of light stimulate the retinas of the eyes, a nerve impulse is sent to a region of the hypothalamus called the suprachiasmatic nucleus (SCN), which is important in regulating biological rhythms. From the SCN, the nerve signal is carried to the spinal cord and eventually to the pineal gland, where the production of melatonin is inhibited. As a result, blood levels of melatonin fall, promoting wakefulness. In contrast, as light levels decline—such as during the evening—melatonin production increases, boosting blood levels and causing drowsiness.
The secretion of melatonin may influence the body’s circadian rhythms, the dark-light fluctuations that affect not only sleepiness and wakefulness, as well as appetite and body temperature. Interestingly, children have higher melatonin levels than adults, which may prevent the release of gonadotropins from the anterior pituitary, thereby inhibiting the onset of puberty. Finally, the antioxidant role of melatonin is the subject of current research.
Jet lag occurs when a person travels across several time zones and feels sleepy during the day or wakeful at night. Traveling across multiple time zones significantly disturbs the light-dark cycle regulated by melatonin. It can take up to several days for melatonin synthesis to adjust to the light-dark patterns in the new environment, resulting in jet lag. Some air travelers take melatonin supplements to induce sleep.
The thymus is an organ of the immune system that is larger and more active during infancy and early childhood, and it begins to atrophy as we age. Its endocrine function is the production of a group of hormones called thymosins that contribute to the development and differentiation of T lymphocytes, which are immune cells. Although the role of thymosins is not yet well understood, it is clear that they contribute to the immune response. Thymosins have been found in tissues other than the thymus and have a wide variety of functions, so the thymosins cannot be strictly categorized as thymic hormones.
A butterfly-shaped organ, the thyroid gland is located anterior to the trachea, just inferior to the larynx. The medial region, called the isthmus, is flanked by wing-shaped left and right lobes. Each of the thyroid lobes is embedded with parathyroid glands, primarily on their posterior surfaces. The tissue of the thyroid gland is composed mostly of thyroid follicles. The follicles are made up of a central cavity filled with a sticky fluid called colloid. Surrounded by a wall of epithelial follicle cells, the colloid is the center of thyroid hormone production, and that production is dependent on the hormones’ essential and unique component: iodine.
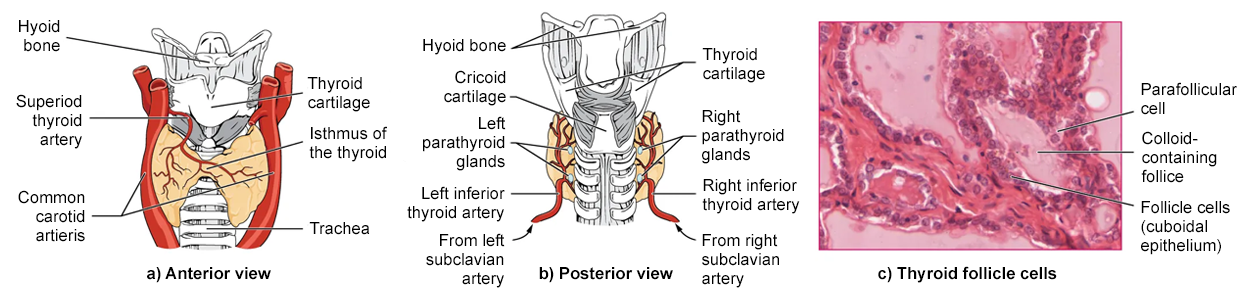
Hormones are produced in the colloid when atoms of the mineral iodine attach to a glycoprotein, called thyroglobulin, that is secreted into the colloid by the follicle cells.
These hormones remain in the colloid center of the thyroid follicles until TSH stimulates endocytosis of colloid back into the follicle cells. There, lysosomal enzymes break apart the thyroglobulin colloid, releasing free T₃ and T₄, which diffuse across the follicle cell membrane and enter the bloodstream.
In the bloodstream, less than one percent of the circulating T₃ and T₄ remains unbound. These free T₃ and T₄ can cross the lipid bilayer of cell membranes and be taken up by cells. The remaining 99 percent of circulating T₃ and T₄ is bound to specialized transport proteins called thyroxine-binding globulins (TBGs), to albumin, or to other plasma proteins. This “packaging” prevents their free diffusion into body cells. When blood levels of T₃ and T₄ begin to decline, bound T₃ and T₄ are released from these plasma proteins and readily cross the membrane of target cells. T₃ is more potent than T₄, and many cells convert T₄ to T₃ through the removal of an iodine atom.
The release of T₃ and T₄ from the thyroid gland is regulated by thyroid-stimulating hormone (TSH). Low blood levels of T₃ and T₄ stimulate the release of thyrotropin-releasing hormone (TRH) from the hypothalamus, which triggers secretion of TSH from the anterior pituitary. In turn, TSH stimulates the thyroid gland to secrete T₃ and T₄. The levels of TRH, TSH, T₃, and T₄ are regulated by a negative feedback system in which increasing levels of T₃ and T₄ decrease the production and secretion of TSH.
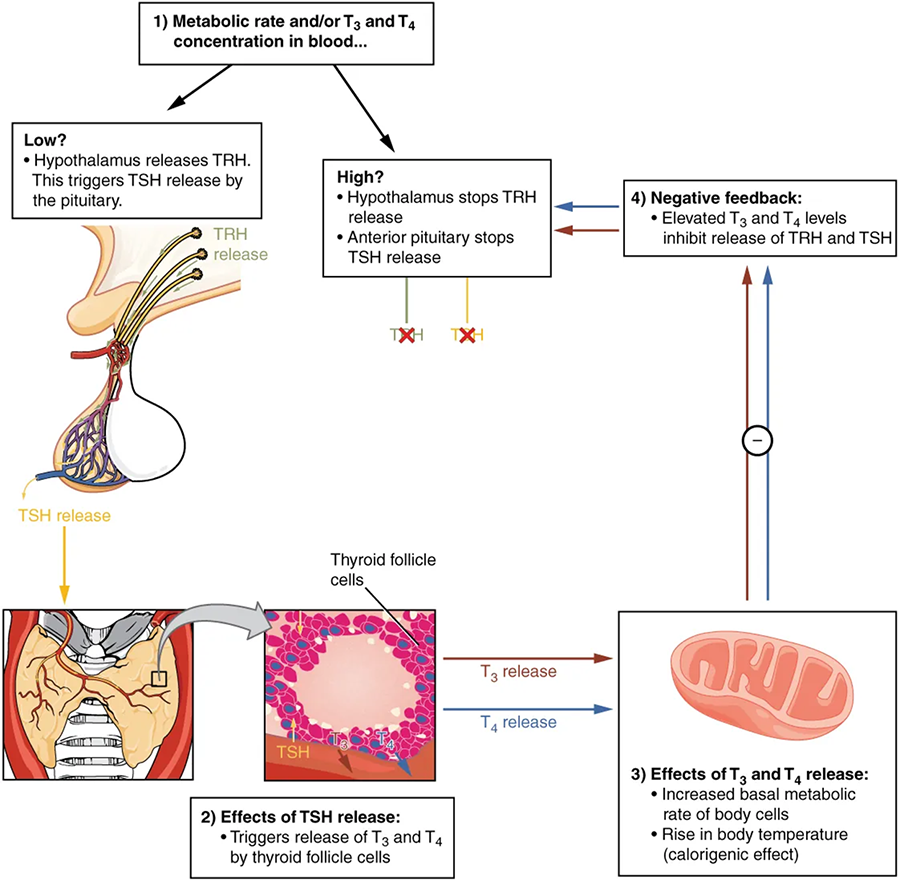
The thyroid hormones, T₃ and T₄, are often referred to as metabolic hormones because their levels influence the body’s basal metabolic rate, the amount of energy used by the body at rest. When T₃ and T₄ bind to intracellular receptors located on the mitochondria, they cause an increase in nutrient breakdown and the use of oxygen to produce ATP. In addition, T₃ and T₄ initiate the transcription of genes involved in glucose oxidation. Although these mechanisms prompt cells to produce more ATP, the process is inefficient, and an abnormally increased level of heat is released as a byproduct of these reactions. This so-called calorigenic effect (calor- = “heat”) raises body temperature.
Adequate levels of thyroid hormones are also required for protein synthesis and for fetal and childhood tissue development and growth. They are especially critical for normal development of the nervous system both in utero and in early childhood, and they continue to support neurological function in adults. As noted earlier, these thyroid hormones have a complex interrelationship with reproductive hormones, and deficiencies can influence libido, fertility, and other aspects of reproductive function. Finally, thyroid hormones increase the body’s sensitivity to catecholamines (epinephrine and norepinephrine) from the adrenal medulla by upregulation of receptors in the blood vessels. When levels of T₃ and T₄ hormones are excessive, this effect accelerates the heart rate, strengthens the heartbeat, and increases blood pressure. Because thyroid hormones regulate metabolism, heat production, protein synthesis, and many other body functions, thyroid disorders can have severe and widespread consequences.
The parathyroid glands are tiny, round structures usually found embedded in the posterior surface of the thyroid gland. A thick connective tissue capsule separates the glands from the thyroid tissue. Most people have four parathyroid glands, but occasionally, there are more in the tissues of the neck or chest. The function of one type of parathyroid cells, the oxyphil cells, is not clear. The primary functional cells of the parathyroid glands are the chief cells. These epithelial cells produce and secrete the parathyroid hormone (PTH), the major hormone involved in the regulation of blood calcium levels.
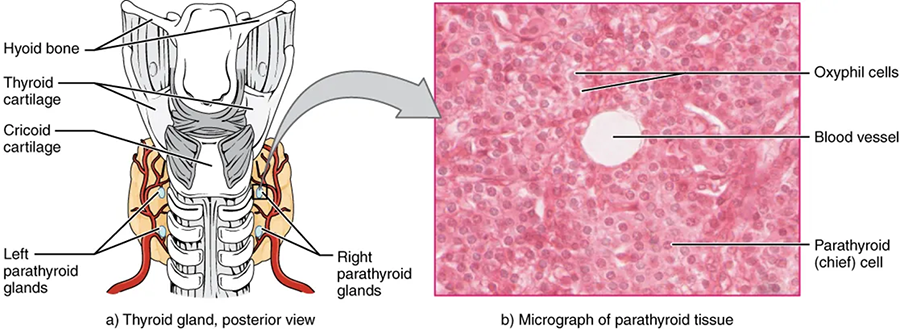
The parathyroid glands produce and secrete PTH, a peptide hormone, in response to low blood calcium levels. PTH secretion causes the release of calcium from the bones by stimulating osteoclasts, which secrete enzymes that degrade bone and release calcium into the interstitial fluid. PTH also inhibits osteoblasts, the cells involved in bone deposition, thereby sparing blood calcium. PTH causes increased reabsorption of calcium (and magnesium) in the kidney tubules from the urine filtrate. In addition, PTH initiates the production of the steroid hormone calcitriol (also known as 1,25-dihydroxyvitamin D), which is the active form of vitamin D₃, in the kidneys. Calcitriol then stimulates increased absorption of dietary calcium by the intestines. A negative feedback loop regulates the levels of PTH, with rising blood calcium levels inhibiting further release of PTH.
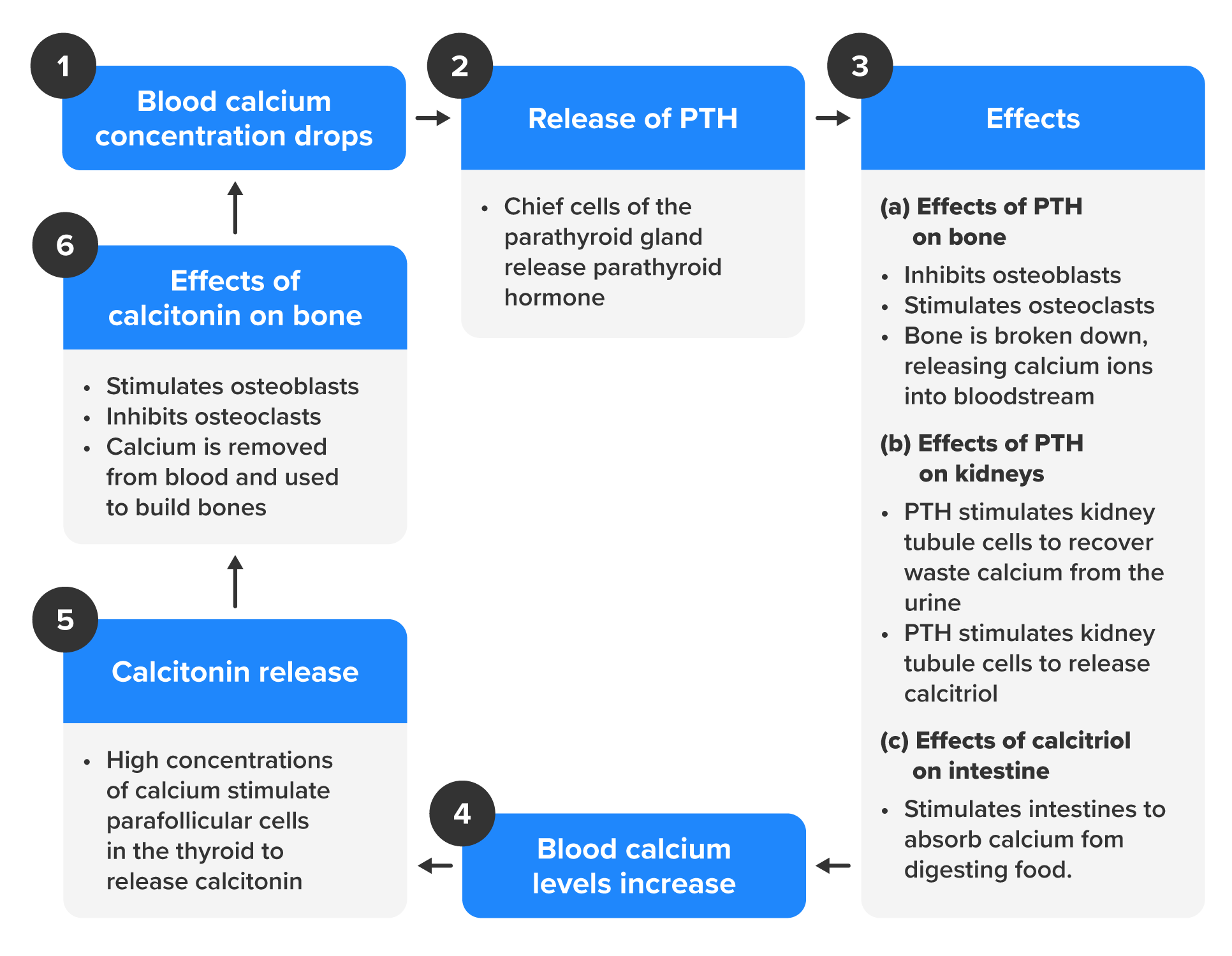
When blood calcium levels are high, calcitonin is produced and secreted by the parafollicular cells of the thyroid gland. As discussed earlier, calcitonin inhibits the activity of osteoclasts, reduces the absorption of dietary calcium in the intestine, and signals the kidneys to reabsorb less calcium, resulting in larger amounts of calcium excreted in the urine.
Source: THIS TUTORIAL HAS BEEN ADAPTED FROM "ANATOMY AND PHYSIOLOGY 2E" AT OpenStax. ACCESS FOR FREE AT https://openstax.org/details/books/anatomy-and-physiology-2e. LICENSING: CREATIVE COMMONS ATTRIBUTION 4.0 INTERNATIONAL.
REFERENCES
Zhu, Y., Verkhratsky, A., Chen, H., & Yi, C. (2025). Understanding glucose metabolism and insulin action at the blood-brain barrier: Implications for brain health and neurodegenerative diseases. Acta Physiologica, 241(2), e14283. doi.org/10.1111/apha.14283