Table of Contents |
Following the somewhat serendipitous discovery of radioactivity by Becquerel, many prominent scientists began to investigate this new, intriguing phenomenon.
During the beginning of the twentieth century, many radioactive substances were discovered, the properties of radiation were investigated and quantified, and a solid understanding of radiation and nuclear decay was developed.
The spontaneous change of an unstable nuclide into another is radioactive decay. The unstable nuclide is called the parent nuclide; the nuclide that results from the decay is known as the daughter nuclide. The daughter nuclide may be stable, or it may decay itself. The radiation produced during radioactive decay is such that the daughter nuclide is more stable than the parent nuclide.
Ernest Rutherford’s experiments involving the interaction of radiation with a magnetic or electric field helped him determine that one type of radiation consisted of positively charged and relatively massive α particles; a second type was made up of negatively charged and much less massive β particles; and a third was uncharged electromagnetic waves, γ rays.
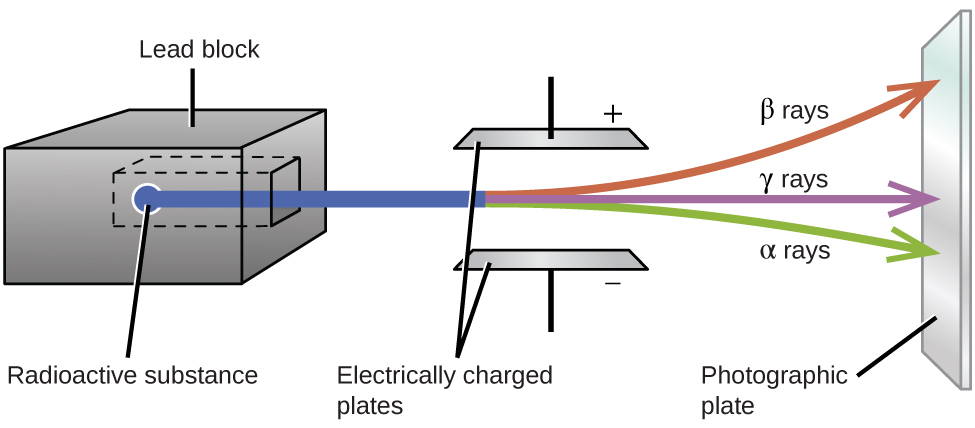
The image above shows a radioactive substance emitting alpha, beta, and gamma rays out of a lead box, which passes through two electrically charged plates onto a photographic plate. The alpha particles, which are attracted to the negative plate and deflected by a relatively small amount, must be positively charged and relatively massive. The beta particles, which are attracted to the positive plate and deflected a relatively large amount, must be negatively charged and relatively light, and the gamma rays, which are unaffected by the electric field, must be uncharged.
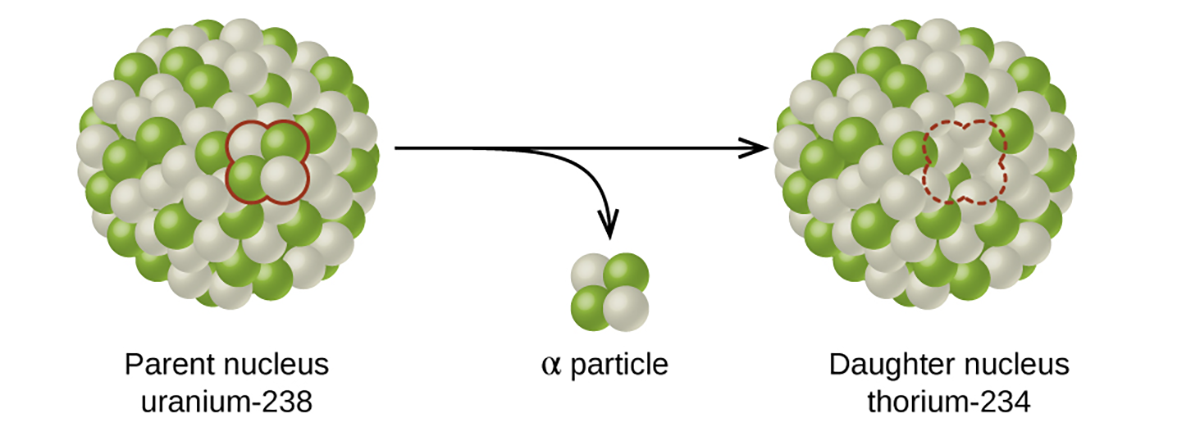
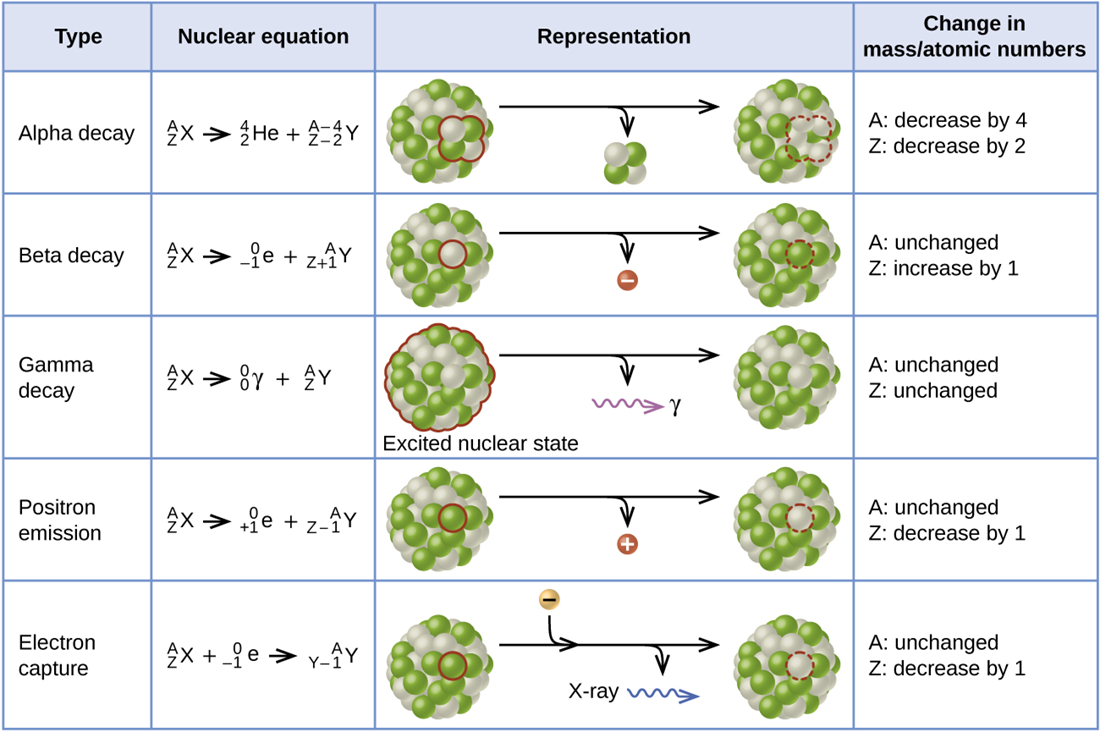
We classify different types of radioactive decay by the radiation produced. Alpha (α) decay is the emission of an α particle from the nucleus. For example, polonium-210 undergoes α decay:
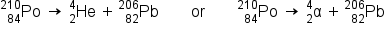
Alpha decay occurs primarily in heavy nuclei (A > 200, Z > 83). Because the loss of an α particle gives a daughter nuclide with a mass number four units smaller and an atomic number two units smaller than those of the parent nuclide, the daughter nuclide has a larger n:p ratio than the parent nuclide.
Beta (β) decay is the emission of an electron from a nucleus. Iodine-131 is an example of a nuclide that undergoes β decay:

Beta-decay, which can be thought of as the conversion of a neutron into a proton and a β particle, is observed in nuclides with a large n:p ratio. The beta particle (electron) emitted is from the atomic nucleus and is not one of the electrons surrounding the nucleus. Emission of an electron does not change the mass number of the nuclide but does increase the number of its protons and decrease the number of its neutrons. Consequently, the n:p ratio is decreased, and the daughter nuclide is more stable than the parent nuclide.
Gamma emission (γ emission) is observed when a nuclide is formed in an excited state and then decays to its ground state with the emission of a γ ray, a quantum of high-energy electromagnetic radiation. The presence of a nucleus in an excited state is often indicated by an asterisk (*). Cobalt-60 emits γ radiation and is used in many applications including cancer treatment:
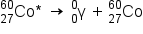
There is no change in mass number or atomic number during the emission of a γ ray unless the γ emission accompanies one of the other modes of decay.
Positron emission (β+ decay) is the emission of a positron from the nucleus. Oxygen-15 is an example of a nuclide that undergoes positron emission:
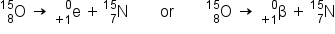
Positron emission is observed for nuclides in which the n:p ratio is low. Positron decay is the conversion of a proton into a neutron with the emission of a positron. The n:p ratio increases and the daughter nuclide is more stable than the parent nuclide.
Electron capture occurs when one of the inner electrons in an atom is captured by the atom’s nucleus. For example, potassium-40 undergoes electron capture:
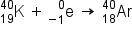
Electron capture occurs when an inner shell electron combines with a proton and is converted into a neutron. The loss of an inner shell electron leaves a vacancy that will be filled by one of the outer electrons. As the outer electron drops into the vacancy, it will emit energy. In most cases, the energy emitted will be in the form of an X-ray.
Like positron emission, electron capture occurs for “proton-rich” nuclei. Electron capture has the same effect on the nucleus as does positron emission: The atomic number is decreased by one and the mass number does not change. This increases the n:p ratio, and the daughter nuclide is more stable than the parent nuclide. Whether electron capture or positron emission occurs is difficult to predict.
A balanced chemical reaction equation reflects the fact that during a chemical reaction, bonds break and form, and atoms are rearranged, but the total numbers of atoms of each element are conserved and do not change. A balanced nuclear reaction equation indicates that there is a rearrangement during a nuclear reaction, but of nucleons (subatomic particles within the atoms’ nuclei) rather than atoms.
If the atomic number and the mass number of all but one of the particles in a nuclear reaction are known, we can identify the particle by balancing the reaction. For instance we could determine that 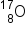 is a product of the nuclear reaction of
is a product of the nuclear reaction of 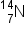 and
and 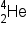 if we knew that a proton,
if we knew that a proton, 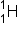 , was one of the two products.
, was one of the two products.
EXAMPLE
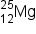 ) produces a proton and a nuclide of another element. Identify the new nuclide produced.
) produces a proton and a nuclide of another element. Identify the new nuclide produced.
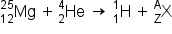
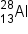 .
.
Follow this link to WebElements to view an accessible version of the periodic table of elements.
IN CONTEXT
Following are the equations of several nuclear reactions that have important roles in the history of nuclear chemistry:
- The first naturally occurring unstable element that was isolated, polonium, was discovered by the Polish scientist Marie Curie and her husband Pierre in 1898. It decays, emitting α particles:
- The first nuclide to be prepared by artificial means was an isotope of oxygen,
O. It was made by Ernest Rutherford in 1919 by bombarding nitrogen atoms with α particles:
- James Chadwick discovered the neutron in 1932, as a previously unknown neutral particle produced along with
C by the nuclear reaction between
Be and
He:
- The first element to be prepared that does not occur naturally on the earth, technetium, was created by bombardment of molybdenum by deuterons (heavy hydrogen,
), by Emilio Segre and Carlo Perrier in 1937:
- The first controlled nuclear chain reaction was carried out in a reactor at the University of Chicago in 1942. One of the many reactions involved was:
Source: THIS TUTORIAL HAS BEEN ADAPTED FROM OPENSTAX “CHEMISTRY: ATOMS FIRST 2E”. ACCESS FOR FREE AT Chemistry: Atoms First 2e. LICENSE: CREATIVE COMMONS ATTRIBUTION 4.0 INTERNATIONAL
REFERENCES
Winter, M. (2021). The periodic table of the elements. The periodic table of the elements by WebElements. www.webelements.com/.