Table of Contents |
In another lesson, you learned about mechanisms of action of antibacterial medications. Because bacteria are prokaryotic, there are many differences between their cells and eukaryotic cells. These differences help us identify useful targets for medications that affect bacterial structures and metabolic processes absent or different enough to be unharmed in eukaryotic cells.
In this lesson, you will learn about ways in which other types of pathogens are targeted by medications and some of the challenges involved in treating each type of pathogen.
Fungi are eukaryotes, meaning that many modes of action of antibacterial medications are not useful in developing antifungal medications. The most common mode of action for antifungal drugs is disruption of the cell membrane due to an important difference between animal and fungal cell membranes.
Animal cell membranes contain cholesterol, but fungal cell membranes contain a closely related compound called ergosterol. Both of these compounds help to maintain appropriate membrane fluidity.
Imidazoles are synthetic fungicides that disrupt ergosterol biosynthesis. These are commonly used to treat fungal skin infections. Triazole drugs, including fluconazole, also inhibit ergosterol biosynthesis and exhibit more selective toxicity than imidazoles. Allylamines, which are structurally different synthetic antifungal medications, inhibit an earlier step in ergosterol biosynthesis.
Polyenes are antifungal agents produced by certain actinomycete soil bacteria and structurally related to macrolides. They bind to ergosterol in fungal cytoplasmic membranes, creating pores. Common examples include nystatin and amphotericin B.
A few antifungal medications have other mechanisms of action. These include echinocandins, which block the synthesis of β(1→3) glucan found in fungal cell walls but not in human cells. Polyoxins and nikkomycins target chitin synthesis, affecting chitin found in fungal cell walls but absent from human cells. Griseofulvin is thought to disrupt formation of the spindle during mitosis, thus inhibiting cell division. Because of its potential for causing toxic effects on the liver, it is sometimes used to treat skin infections when other treatments are ineffective.
There are also a few drugs that act as antimetabolites against fungal processes. Atovaquinone (a member of the naphthoquinone drug class) is a semisynthetic antimetabolite for the fungal and protozoal versions of a mitochondrial cytochrome important in electron transport.
The image below summarizes some mechanisms of action of antifungal medications, which are also summarized in detail in the table below.
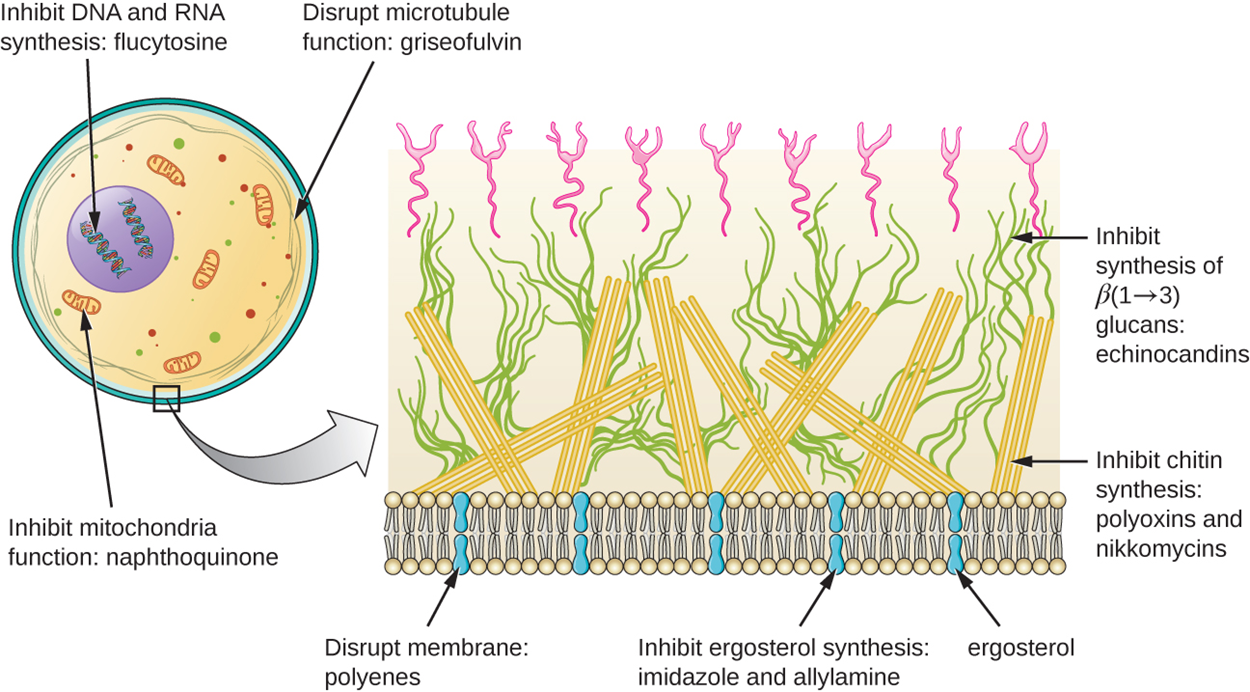
| Common Antifungal Drugs | |||
|---|---|---|---|
| Mechanism of Action | Drug Class | Specific Drugs | Clinical Uses |
| Inhibit ergosterol synthesis | Imidazoles | Miconazole, ketoconazole, clotrimazole | Fungal skin infections and vaginal yeast infections |
| Triazoles | Fluconazole | Systemic yeast infections, oral thrush, and cryptococcal meningitis | |
| Allylamines | Terbinafine | Dermatophytic skin infections (athlete’s foot, ring worm, jock itch), and infections of fingernails and toenails | |
| Bind ergosterol in the cell membrane and create pores that disrupt the membrane | Polyenes | Nystatin | Used topically for yeast infections of skin, mouth, and vagina; also used for fungal infections of the intestine |
| Amphotericin B | Various systemic fungal infections | ||
| Inhibit cell wall synthesis | Echinocandins | Caspofungin | Aspergillosis and systemic yeast infections |
| Not applicable | Nikkomycin Z | Coccidioidomycosis (Valley fever) and yeast infections | |
| Inhibit microtubules and cell division | Not applicable | Griseofulvin | Dermatophytic skin infections |
Protozoans are also eukaryotes, adding to the challenge of developing appropriate treatments. Some antiprotozoal medications are antimetabolites (e.g., atovaquone, proguanil, and artemisinins). For example, malaria and toxoplasmosis can be treated using a synthetic sulfa drug called sulfadiazine that competitively inhibits an enzyme in folic acid production.
Two classes of antiprotozoal drugs interfere with nucleic acid synthesis. Nitroimidazoles can induce DNA strand breakage in low-oxygen environments, interfering with DNA replication. However, one of these medications (metronidazole) is associated with the risk of cancer development in humans. Quinolones are thought to interfere with heme detoxification, which is needed to break down hemoglobin in red blood cells into amino acids. The synthetic derivatives chloroquine, quinacrine, and mefloquine are commonly used as antimalarials. However, long-term use of chloroquine or mefloquine may cause serious side effects.
Pentamidine is another type of synthetic antiprotozoal drug that is thought to interfere with DNA replication.
The table below summarizes antiprotozoal medications.
| Common Antiprotozoal Drugs | |||
|---|---|---|---|
| Mechanism of Action | Drug Class | Specific Drugs | Clinical Uses |
| Inhibit electron transport in mitochondria | Naphthoquinone | Antovaquinone | Malaria, babesiosis, and toxoplasmosis |
| Inhibit folic acid synthesis | Not applicable | Proguanil | Combination therapy with atovaquone for malaria treatment and prevention |
| Sulfonamide | Sulfadiazine | Malaria and toxoplasmosis | |
| Not applicable | Pyrimethamine | Combination therapy with sulfadoxine (sulfa drug) for malaria | |
| Produces damaging reactive oxygen species | Not applicable | Artemisinin | Combination therapy to treat malaria |
| Inhibit DNA synthesis | Nitroimidazoles | Metronidazole, tinidazole | Infections caused by Giardia lamblia, Entamoeba histolytica, and Trichomonas vaginalis |
| Not applicable | Pentamidine | African sleeping sickness and leishmaniasis | |
| Inhibit heme detoxification | Quinolines | Chloroquine | Malaria and infections with E. histolytica |
| Mepacrine, mefloquine | Malaria | ||
Helminths are multicellular eukaryotic worms, but several effective classes of antihelminthic drugs have been developed.
Synthetic benzimidazoles (e.g., mebendazole and albendazole) bind to helminthic β-tubulin, which is part of the cytoskeleton required for microtubule formation. These medications are also effective against many protozoans, fungi, and viruses.
Avermectins are another antihelminthic class. The semisynthetic derivative ivermectin binds to glutamate-gated chloride channels specific to invertebrates including helminths. This blocks neuronal transmission. Ivermectin is used to treat roundworm diseases and to target some parasitic insects.
EXAMPLE
You may be familiar with ivermectin because it is commonly used in veterinary medicine. During the COVID-19 pandemic, there was considerable discussion of ivermectin as a potential treatment. The Food and Drug Administration (FDA) specifically warned against taking ivermectin to treat COVID-19, warning that it has never been approved for this purpose (FDA, 2021).Niclosamide has been used to treat tapeworm infections for over 50 years and appears to have two mechanisms: inhibition of ATP formation under anaerobic conditions and inhibition of oxidative phosphorylation in mitochondria. It is not absorbed from the gastrointestinal tract, so it can target gastrointestinal parasites. It also appears to be effective against some other pathogens.
Praziquantel is a synthetic antihelminthic medication used to treat parasitic tapeworms, liver flukes, and schistosomiasis (caused by blood flukes). Its mode of action is unclear, but it appears to cause an influx of calcium into worms.
Thioxanthones inhibit RNA synthesis, but can have some serious adverse effects. The first thioxanthone, lucanthone, is no longer used because it has such a high risk of causing neurological, cardiovascular, gastrointestinal, and liver problems. Oxamniquine, which is less toxic, is only effective against one species. However, increased evidence of resistance to praziquantel has increased interest in further research into these compounds and their derivatives.
The table below summarizes important antihelminthic medications.
| Common Antihelminthic Drugs | |||
|---|---|---|---|
| Mechanism of Action | Drug Class | Specific Drugs | Clinical Uses |
| Inhibit microtubule formation, reducing glucose uptake | Benzimidazoles | Mebendazole, albendazole | Variety of helminth infections |
| Block neuronal transmission, causing paralysis and starvation | Avermectins | Ivermectin | Roundworm diseases, including river blindness and strongyloidiasis, and treatment of parasitic insects |
| Inhibit ATP production | Not applicable | Niclosamide | Intestinal tapeworm infections |
| Induce calcium influx | Not applicable | Praziquantel | Schistosomiasis (blood flukes) |
Antiviral medications have been difficult to develop because viruses use host cell machinery to produce new viruses. Viral structure is relatively simple. However, multiple types of antivirals are now available.
Many antiviral drugs are nucleoside analogs that function by inhibiting nucleic acid biosynthesis. These include acyclovir, which requires activation by a viral enzyme and has an increased affinity for the active form of viral DNA polymerase versus host cell DNA polymerase.
Ribavirin appears to interfere with DNA and RNA synthesis, but its mode of action is not entirely clear. It also appears to inhibit the RNA polymerase of the hepatitis C virus.
Amantadine and rimantadine are medications that bind to a transmembrane protein involved in the escape of the influenza virus from endosomes. This prevents viral RNA release into host cells, inhibiting viral replication. Unfortunately, resistance to these medications is becoming increasingly common.
Neuraminidase inhibitors target influenza viruses by blocking the activity of influenza virus neuraminidase, which prevents the release of the virus from infected cells. The three types currently in use are oseltamivir, zanamivir, and peramivir. They decrease flu symptoms and shorten the duration of the illness. Oseltamivir is most commonly used as it is administered orally, whereas zanamivir is inhaled and peramivir is administered intravenously.
The images below illustrate the activity of several types of antivirals. The first image shows the mechanism of acyclovir.
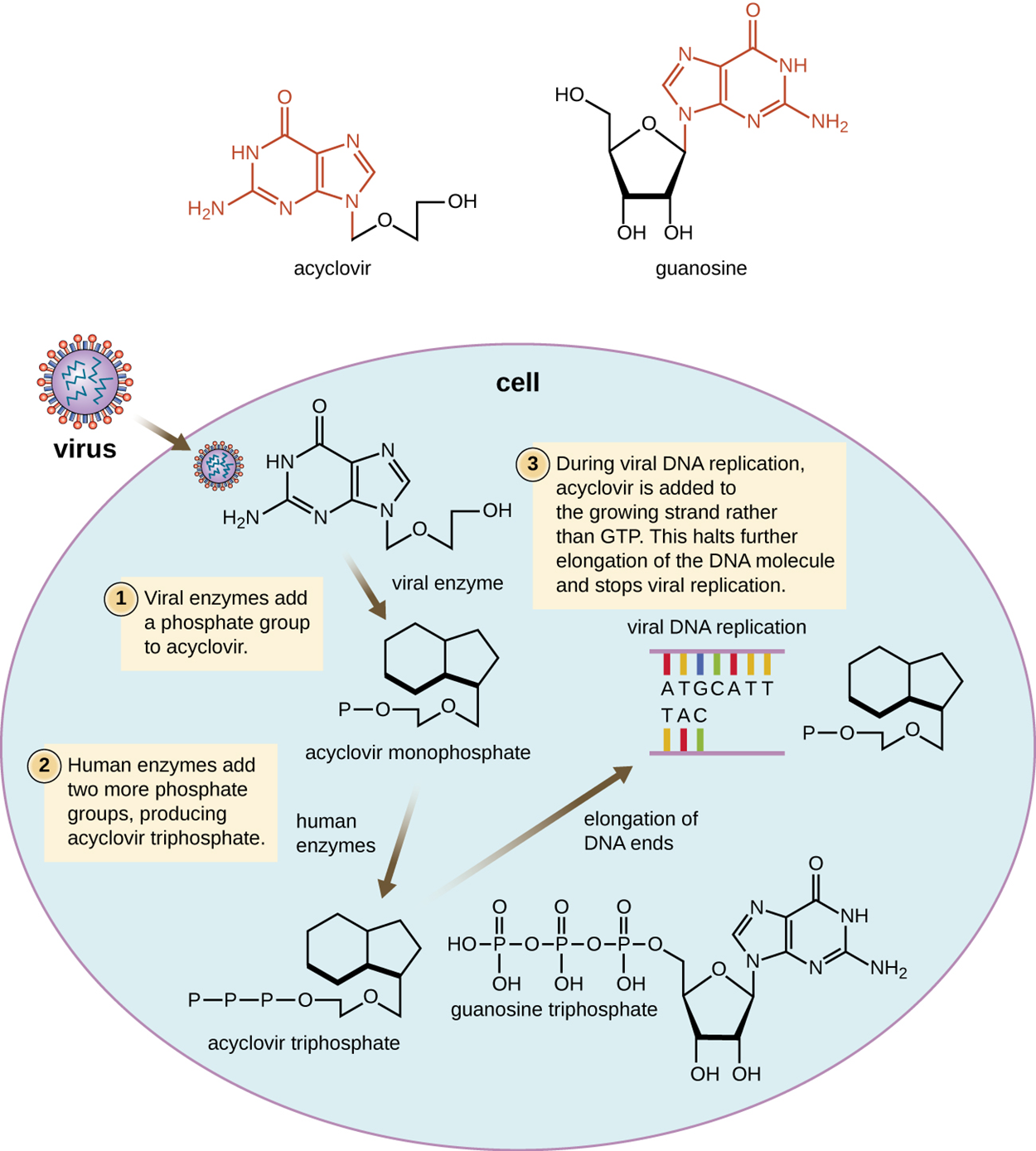
The image below illustrates how different drugs target different parts of the HIV life cycle.
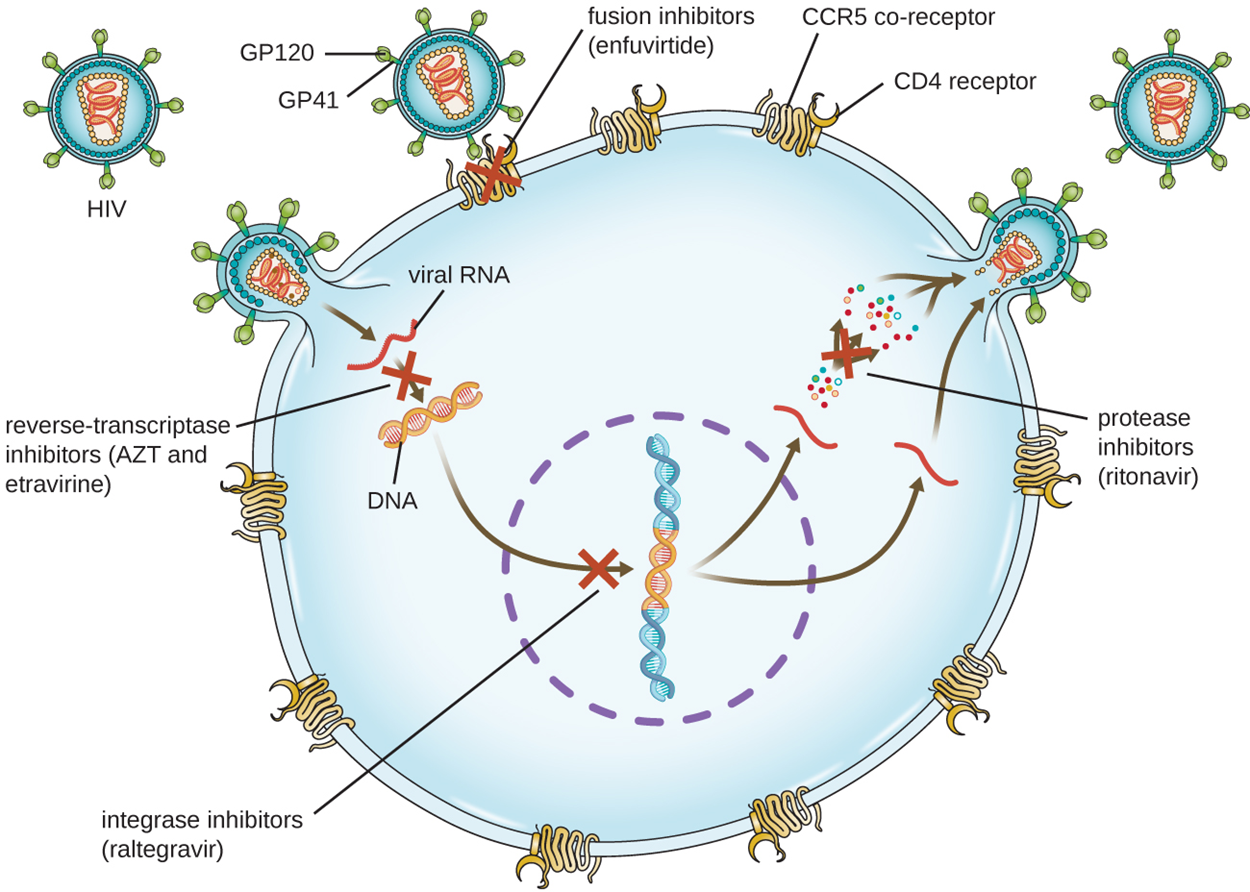
The table below gives examples of the mechanisms of action and uses of common antiviral drugs.
| Common Antiviral Drugs | ||
|---|---|---|
| Mechanism of Action | Drug | Clinical Uses |
| Nucleoside analog inhibition of nucleic acid synthesis | Acyclovir | Herpes virus infections |
| Azidothymidine/zidovudine (AZT) | HIV infections | |
| Ribavirin | Hepatitis C virus and respiratory syncytial virus infections | |
| Vidarabine | Herpes virus infections | |
| Sofosbuvir | Hepatitis C virus infections | |
| Non-nucleoside noncompetitive inhibition | Etravirine | HIV infections |
| Inhibit escape of virus from endosomes | Amantadine, rimantadine | Infections with influenza virus |
| Inhibit neuraminadase | Oseltamivir, zanamivir, peramivir | Infections with influenza virus |
| Inhibit viral uncoating | Pleconaril | Serious enterovirus infections |
| Inhibition of protease | Ritonavir | HIV infections |
| Simeprevir | Hepatitis C virus infections | |
| Inhibition of integrase | Raltegravir | HIV infections |
| Inhibition of membrane fusion | Enfuvirtide | HIV infections |
Source: THIS TUTORIAL HAS BEEN ADAPTED FROM OPENSTAX “MICROBIOLOGY.” ACCESS FOR FREE AT openstax.org/details/books/microbiology. LICENSE: CC ATTRIBUTION 4.0 INTERNATIONAL.
REFERENCES
FDA, FAQ: COVID-19 and Ivermectin Intended for Animals. (2022, October, 13). Retrieved November 16, 2022, from
www.fda.gov/animal-veterinary/product-safety-information/faq-covid-19-and-ivermectin-intended-animals