Table of Contents |
A lipid is one of a highly diverse group of organic compounds made up mostly of hydrocarbons. Recall that a hydrocarbon is a carbon skeleton (the carbon “framework”) with many covalently bonded hydrogen atoms. The few oxygen atoms they contain are often at the periphery of the molecule. Their nonpolar hydrocarbons make all lipids hydrophobic (not dissolved in water), at least in part. Recall that hydrophobic means water (hydro)-fearing (phobic).
Four general groups of lipids exist based on their chemical structure and function: triglycerides, phospholipids, steroids, and prostaglandins.
A triglyceride is one of the most common dietary lipid groups, and the type found most abundantly in body tissues. This compound, which is commonly referred to as fat, is formed from the synthesis of two types of molecules.
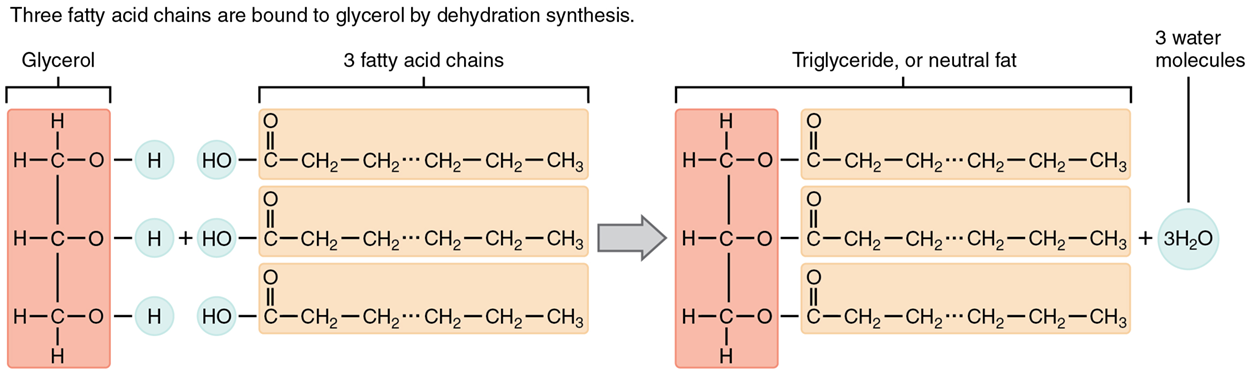
The carbon skeleton of fatty acids can vary in length from two to more than twenty-two carbon atoms. All fatty acids are classified based on the bonding structure between these carbon atoms, but you may be familiar with them from nutrition labels and their effects on health.
Saturated fats are a type of fat that only contain single covalent bonds in the fatty acid tail. Saturated fats are often animal fats. Because there are no double bonds, the fatty acid side chains are fairly straight and therefore can pack together neatly. This dense packing is the reason saturated fats tend to be solid at room temperature.
EXAMPLE
Butter and lard are saturated fats.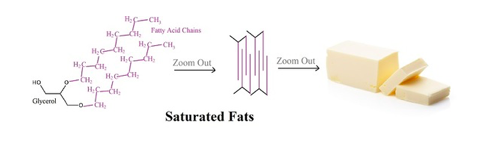
Unsaturated fats are a type of fat that has one or more double covalent bonds between carbons, which are the backbone of the fatty acid. Unsaturated fats are generally more plant-based, whereas saturated fats are more animal-based. The double bond puts a kink in the fatty acid chain, so unsaturated fats can't pack together very tightly. Because of this less dense packing, unsaturated fats tend to be liquid at room temperature.
EXAMPLE
Olive oil is an unsaturated fat.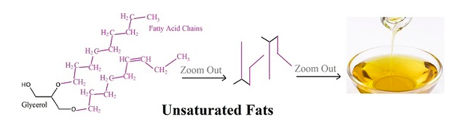
Whereas a diet high in saturated fatty acids increases the risk of heart disease, a diet high in unsaturated fatty acids is thought to reduce the risk. The body can produce most unsaturated fatty acids when necessary. However, there are two fatty acids the body cannot produce and are therefore essential to the human diet, called essential fatty acids. These are omega-3 and omega-6 unsaturated fatty acids.
These fatty acids have their first carbon double bond at the third and sixth hydrocarbon from the methyl group (referred to as the omega end of the molecule), respectively. Omega-3 unsaturated fatty acids are found in cold-water fish, such as salmon, as well as plant-based foods such as flaxseed, soy foods, and walnuts.
Omega-6 unsaturated fatty acids are found in most fried foods since they include soybean, cottonseed, and/or corn oil, as well as in food sources such as walnuts, pine nuts, and sunflower seeds.
Another type of unsaturated fat is something called polyunsaturated fat. The prefix "poly" means many; this type of unsaturated fat could have multiple double bonds. Plant oils, such as olive oil, typically contain both mono- and polyunsaturated fatty acids.
Finally, trans fatty acids found in some processed foods, including some stick and tub margarine, are created artificially by straightening the carbon double bond in unsaturated fatty acids (such as corn oil) so that they can pack tightly together and be solid at room temperature. Like other unsaturated fats, trans fats have a double bond between at least two of the carbons ("C" in the pictures above). Unlike other unsaturated fats, this double bond doesn't put a kink in the chain. The fatty acid chains remain fairly straight and pack densely like saturated fats.
As a group, triglycerides are a major fuel source for the body. You already know that triglycerides can be broken down to provide three fatty acids and one glycerol. These components can be broken down to produce all the required energy for slow physical activity (such as gardening or hiking) and contribute a modest percentage of energy for vigorous physical activity. When excess fatty acids are available in the body from either diet or chemical conversion, they can be combined with a glycerol molecule and stored as triglycerides in fat (adipose) tissue.
Dietary fat also assists the absorption and transport of the nonpolar fat-soluble vitamins A, D, E, and K. Additionally, stored body fat protects and cushions the body’s bones and internal organs, and acts as insulation to retain body heat.
In addition to triglycerides, there are other lipids that are also essential to human functioning, including phospholipids, sterols, and prostaglandins.
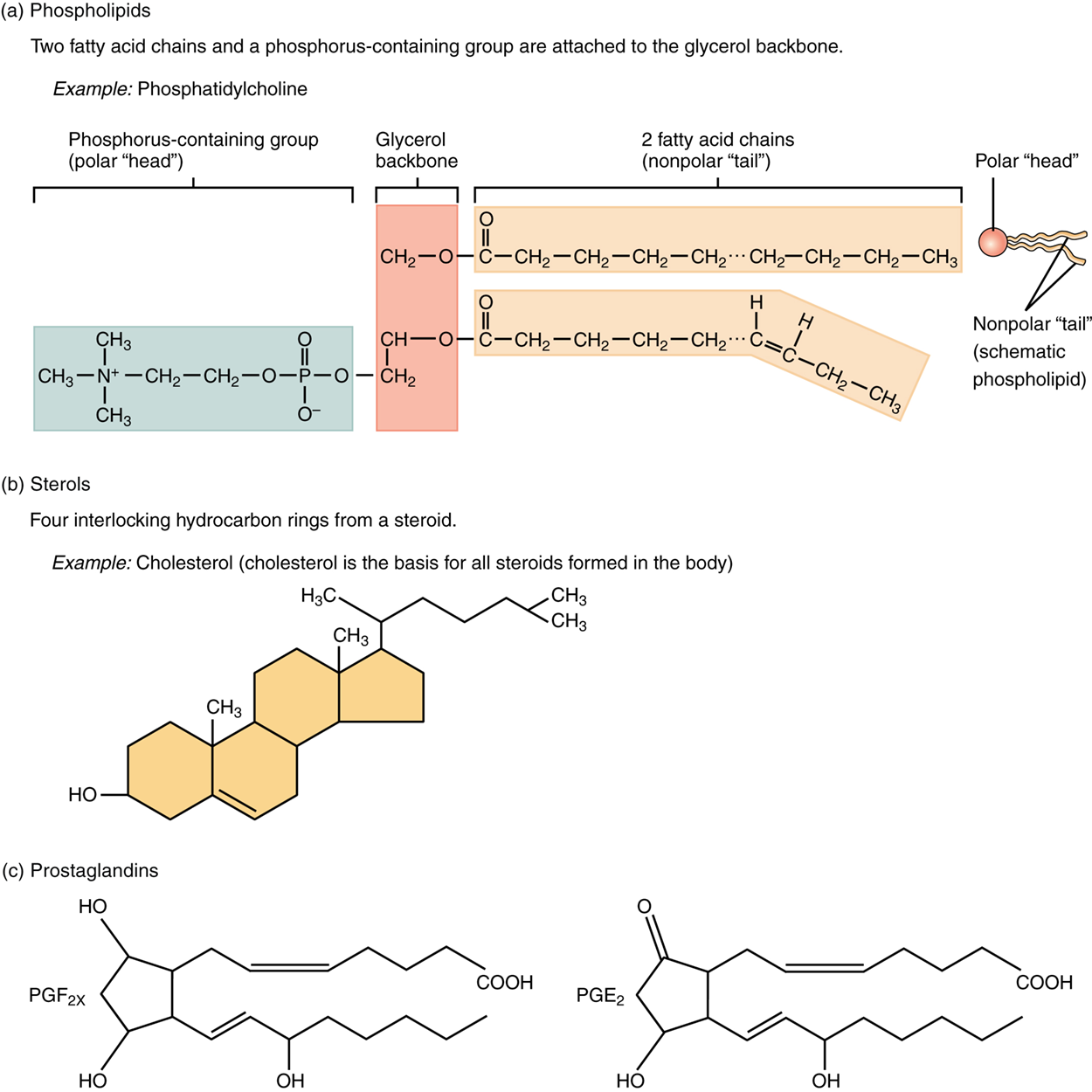
Phospholipids are quite similar in structure to triglycerides but incorporate a molecule of phosphorus into their chemical structure. Whereas triglycerides contain a glycerol molecule and three fatty acids, a phospholipid has a glycerol molecule, two fatty acids, and a phosphate group. You will see in a future lesson that the opposing ends of phospholipids play an important role in the structure and function of every cell in your (or any) body.
A steroid compound, referred to as a sterol, has as its foundation a set of four hydrocarbon rings bonded to a variety of other atoms and molecules. Sterols play roles in physiological activities and maintaining cell membranes. Although both plants and animals synthesize sterols, the type that makes the most important contribution to human structure and function is cholesterol, which is synthesized by the liver in humans and animals and is also present in most animal-based foods. Like other lipids, cholesterol’s hydrocarbon rings make it hydrophobic; however, it has a polar hydroxyl head that is hydrophilic.
Prostaglandins are signaling molecules, similar to hormones. However, while hormones are derived from cholesterol and steroids, prostaglandins are derived from unsaturated fatty acids.
IN CONTEXT
One reason that the omega-3 fatty acids found in fish are beneficial is that they stimulate the production of certain prostaglandins that help regulate aspects of blood pressure and inflammation and thereby reduce the risk for heart disease.
Prostaglandins also sensitize nerves to pain. One class of pain-relieving medications called nonsteroidal anti-inflammatory drugs (NSAIDs) works by reducing the effects of prostaglandins.
SOURCE: THIS TUTORIAL HAS BEEN ADAPTED FROM OPENSTAX “ANATOMY AND PHYSIOLOGY 2E”. ACCESS FOR FREE AT OPENSTAX.ORG/BOOKS/ANATOMY-AND-PHYSIOLOGY-2E/PAGES/1-INTRODUCTION. LICENSE: CREATIVE COMMONS ATTRIBUTION 4.0 INTERNATIONAL.