Table of Contents |
A major part of understanding chemistry is to be aware of how the macroscopic and microscopic domains relate and how chemists model them in the symbolic domain. Recall that the macroscopic domain is the realm of everyday things that are large enough to be sensed directly by human sight or touch. The microscopic domain of chemistry is where all of the real chemistry is taking place. Most of the subjects in the microscopic domain of chemistry are too small to be seen, even with the most advanced microscopes, and may only be pictured in the mind.
Because we cannot see the microscopic domain, we create models that give us a picture or symbolic notation of what is occurring. See the figure below for an example of macroscopic, microscopic, and symbolic domains.
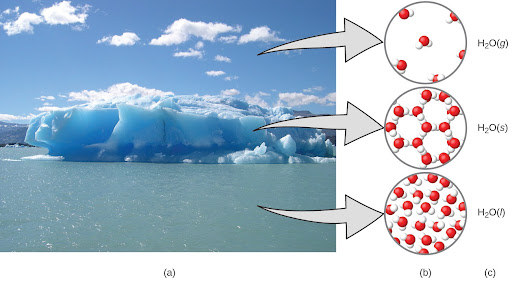
The image above shows moisture in the air, icebergs, and the ocean, all representing water in the macroscopic domain (a). At the molecular level (the microscopic domain), gas molecules are far apart and disorganized, solid water molecules are close together and organized, and liquid molecules are close together and disorganized (b). The formula H2O symbolizes water, and (g), (s), and (l) symbolize its phases (c). Note that clouds are actually comprised of either very small liquid water droplets or solid water crystals. Gaseous water in our atmosphere is not visible to the naked eye, although it may be sensed as humidity.
The symbolic domain contains the specialized language used to represent components of the macroscopic and microscopic domains. Chemical symbols (such as those used in the periodic table), chemical formulas, and chemical equations are part of the symbolic domain, as are graphs, drawings, and calculations. These symbols play an important role in chemistry because they help interpret the behavior of the macroscopic domain in terms of the components of the microscopic domain.
When substances interact (in the microscopic domain), there is often a chemical change that occurs between those substances. It is important to have an accurate and quick shorthand symbolic notation to explain those changes. We call this a chemical reaction, which is a chemical process where one or more reactants are converted into different substances (products).
Because chemists are faced with a wide range of varied interactions between chemical substances, they have found it convenient (or even necessary) to classify chemical interactions by identifying common patterns of reactivity. This lesson provides an introduction to three of the most prevalent types of chemical reactions: precipitation, acid-base, and oxidation-reduction.
A precipitation reaction is one in which dissolved substances react to form one (or more) solid products. Many reactions of this type involve the exchange of ions between ionic compounds in aqueous solution and are sometimes referred to as double displacement, double replacement, or metathesis reactions.
The extent to which a substance may be dissolved in water, or any solvent, is quantitatively expressed as its solubility, defined as the maximum concentration of a substance that can be achieved under specified conditions. Substances with relatively large solubilities are said to be soluble. A substance will precipitate when solution conditions are such that its concentration exceeds its solubility. Substances with relatively low solubilities are said to be insoluble, and these are the substances that readily precipitate from a solution. The table below summarizes the basic solubility rules of various ions found in ionic compounds.
| Soluble Ionic Compounds | Contain these ions: | Exceptions: |
|---|---|---|
|
NH4+ Group I cations: Li+ Na+ K+ Rb+ Cs+ |
none | |
|
Cl- Br- I- |
compounds with Ag+, Hg22+, and Pb2+ | |
| F- | compounds with group 2 metal cations, Pb2+, and Fe3+ | |
|
C2H3O2- HCO3- NO3- ClO3- |
none | |
| SO42- | compounds with Ag+, Ba2+, Ca2+, Hg22+, Pb2+ and Sr2+ | |
| Insoluble Ionic Compounds | Contain these ions: | Exceptions: |
|
CO32- CrO42- PO43- S2- |
compounds with group 1 cations and NH4>+ | |
| OH- | compounds with group 1 cations and Ba2+ |
EXAMPLE
Precipitation is observed when solutions of potassium iodide and lead nitrate are mixed, resulting in the formation of solid lead iodide. The symbolic notation for this macroscopic observation is the chemical reaction shown below:This observation is consistent with the solubility guidelines: The only insoluble compound among all those involved is lead iodide, one of the exceptions to the general solubility of iodide salts.

The solubility guidelines in the table above may be used to predict whether a precipitation reaction will occur when solutions of soluble ionic compounds are mixed together. One merely needs to identify all the ions present in the solution and then consider if a possible cation/anion pairing could result in an insoluble compound.
EXAMPLE
Mixing solutions of silver nitrate and sodium fluoride will yield a solution containing Ag+, NO3-, Na+, and F- ions. Aside from the two ionic compounds originally present in the solutions, AgNO3 and NaF, two additional ionic compounds may be derived from this collection of ions: NaNO3 and AgF. The solubility guidelines indicate all nitrate salts are soluble but that AgF is one of the exceptions to the general solubility of fluoride salts. A precipitation reaction, therefore, is predicted to occur, as described by the following equations:An acid-base reaction is one in which a hydrogen ion, H+, is transferred from one chemical species to another. An acid is a substance that will dissolve in water to yield hydronium ions, H3O+.
EXAMPLE
Consider the equation shown here: HCl(𝑎𝑞) + H2O(𝑎𝑞)⟶Cl−(𝑎𝑞) + H3O+(𝑎𝑞)The process represented by this equation confirms that hydrogen chloride is an acid. When dissolved in water, H3O+ ions are produced by a chemical reaction in which H+ ions are transferred from HCl molecules to H2O molecules.
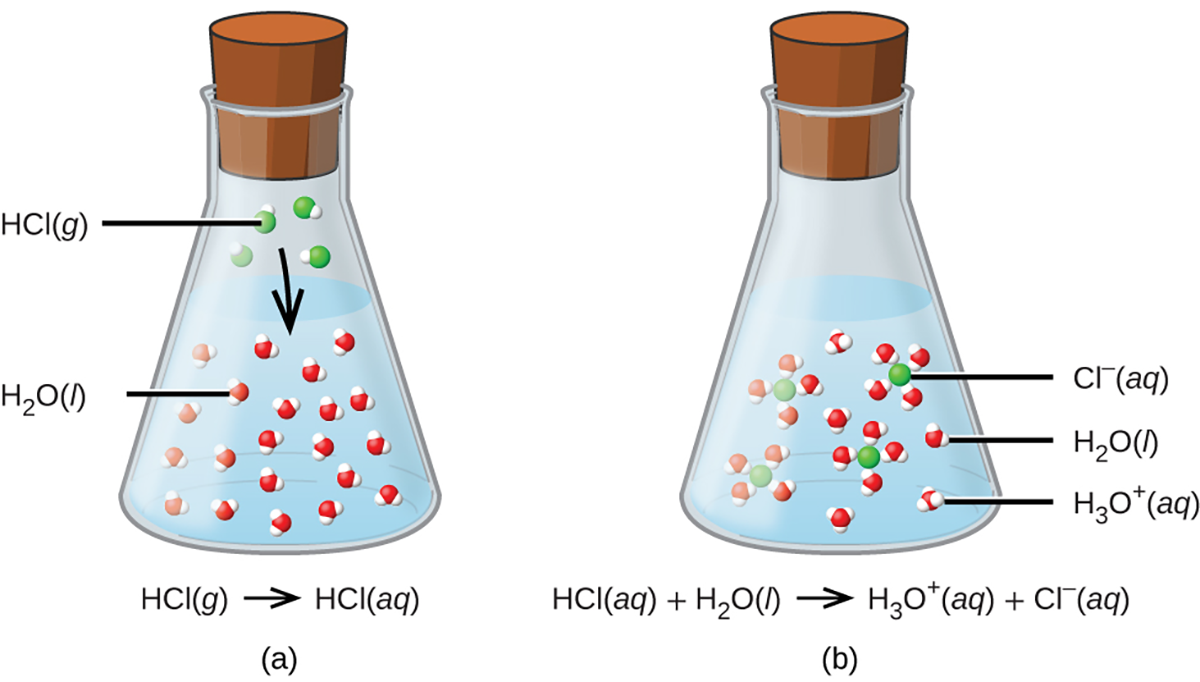
In the image above hydrogen chloride gas dissolves in water, (a) it reacts as an acid, transferring protons to water molecules to yield (b) hydronium ions (and solvated chloride ions). The molecules in the flask represent the microscopic domain and the chemical equation represents the symbolic domain.
Virtually every HCl molecule that dissolves in water will undergo this reaction. Acids that completely react in this fashion are called strong acids, and HCl is one among just a handful of common acid compounds that are classified as strong.
| Compound Formula | Name in Aqueous Solution |
|---|---|
| HBr | hydrobromic acid |
| HCl | hydrochloric acid |
| HI | hydroiodic acid |
| HNO3 | nitric acid |
| HClO4 | perchloric acid |
| H2SO4 | sulfuric acid |
A far greater number of compounds behave as weak acids and only partially react with water, leaving a large majority of dissolved molecules in their original form and generating a relatively small amount of hydronium ions. A familiar example of a weak acid is acetic acid, the main ingredient in food vinegar. When dissolved in water under typical conditions, only about 1% of acetic acid molecules are present in the ionized form, CH3CO2-(aq).
NOTE: The use of a double-arrow in the equation below denotes the partial reaction aspect of this process, a concept addressed fully in another lesson.
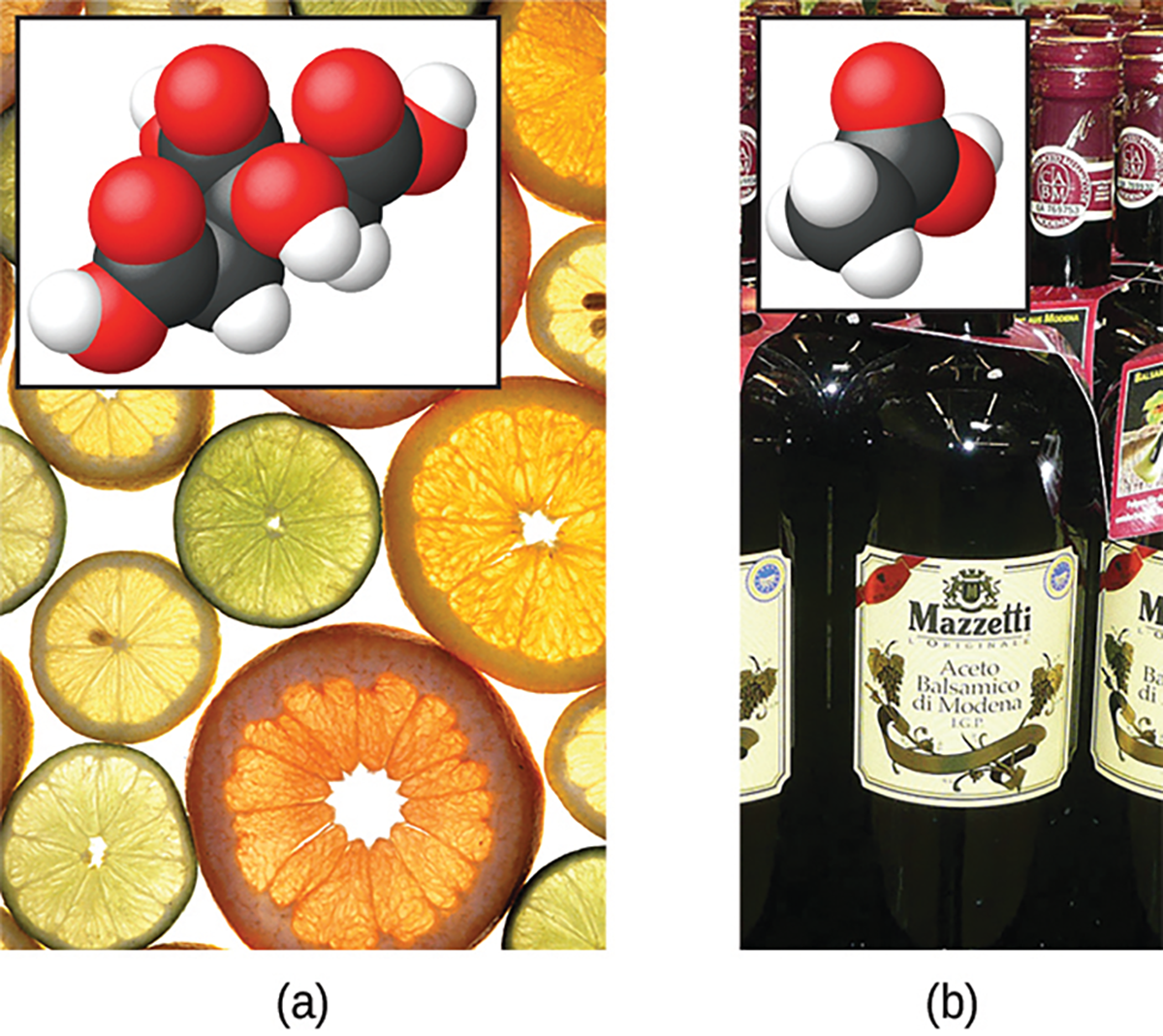
In the above image, fruits such as oranges, lemons, and grapefruit (a) contain the weak acid citric acid, while vinegar (b) contains the weak acid acetic acid.
A base is a substance that will dissolve in water to yield hydroxide ions, OH-. The most common bases are ionic compounds composed of alkali or alkaline earth metal cations (groups 1 and 2) combined with the hydroxide ion, such as NaOH and Ca(OH)2. Unlike the acid compounds discussed previously, these compounds do not react chemically with water; instead, they dissolve and dissociate, releasing hydroxide ions directly into the solution.
EXAMPLE
KOH and Ba(OH)2 dissolve in water and dissociate completely to produce cations (K+ and Ba2+, respectively) and hydroxide ions, OH-. These bases, along with other hydroxides that completely dissociate in water, are considered strong bases. Consider as an example the dissolution of lye (sodium hydroxide) in water:This equation confirms that sodium hydroxide is a base. When dissolved in water, NaOH dissociates to yield Na+ and OH- ions. This is also true for any other ionic compound containing hydroxide ions. Since the dissociation process is essentially complete when ionic compounds dissolve in water under typical conditions, NaOH and other ionic hydroxides are all classified as strong bases.
Unlike ionic hydroxides, some compounds produce hydroxide ions when dissolved by chemically reacting with water molecules. In all cases, these compounds react only partially and so are classified as weak bases. When dissolved in water, ammonia reacts partially to yield hydroxide ions, as shown here:
This is, by definition, an acid-base reaction, in this case involving the transfer of H+ ions from water molecules to ammonia molecules. Under typical conditions, only about 1% of the dissolved ammonia is present as NH4+ ions.
A neutralization reaction is a specific type of acid-base reaction in which the reactants are an acid and a base (but not water), and the products are often a salt and water. A salt is an ionic compound that forms when the negative ion from an acid combines with the positive ion from a base.
EXAMPLE
To illustrate a neutralization reaction, consider what happens when a typical antacid such as milk of magnesia (an aqueous suspension of solid Mg(OH)2) is ingested to ease symptoms associated with excess stomach acid (HCl). Note that in addition to water, this reaction produces a salt, magnesium chloride.The third group of reactions is oxidation-reduction (redox) reactions. Most redox reactions involve the transfer of electrons between reactant species to yield ionic products, such as the reaction between sodium and chlorine to yield sodium chloride:
It is helpful to view the process with regard to each individual reactant in the form of an equation called a half-reaction, which is either the oxidation reaction or the reduction reaction of the redox reaction.
These equations show that Na atoms lose electrons while Cl atoms (in the Cl2 molecule) gain electrons. For redox reactions of this sort, the loss and gain of electrons define the complementary processes that occur:
In this reaction, then, sodium is oxidized and chlorine undergoes reduction. Viewed from a more active perspective, sodium functions as a reducing agent (reductant). Likewise, chlorine functions as an oxidizing agent (oxidant), as it effectively removes electrons from (oxidizes) sodium.
Some redox processes, however, do not involve the transfer of electrons. Consider, for example, a reaction similar to the one yielding NaCl:
The product of this reaction is a covalent compound, so transfer of electrons in the explicit sense is not involved. To clarify the similarity of this reaction to the previous one and permit an unambiguous definition of redox reactions, a property called oxidation number has been defined. The oxidation number (or oxidation state) of an element in a compound is the charge its atoms would possess if the compound was ionic.
IN CONTEXT
Note: The proper convention for reporting charge is to write the number first, followed by the sign (e.g., 2+), while oxidation number is written with the reversed sequence, sign followed by number (e.g., +2). This convention aims to emphasize the distinction between these two related properties.
Using the oxidation number concept, an all-inclusive definition of redox reaction has been established. Oxidation-reduction (redox) reactions are those in which one or more elements involved undergo a change in oxidation number. While the vast majority of redox reactions involve changes in oxidation number for two or more elements, a few interesting exceptions to this rule do exist. Definitions for the complementary processes of this reaction class are correspondingly revised as shown here:
Several subclasses of redox reactions are recognized, including combustion reactions in which the reductant (also called a fuel) and oxidant (often, but not necessarily, molecular oxygen) react vigorously and produce significant amounts of heat, and often light, in the form of a flame.
Single-displacement (replacement) reactions are redox reactions in which an ion in solution is displaced (or replaced) via the oxidation of a metallic element. One common example of this type of reaction is the acid oxidation of certain metals:
Metallic elements may also be oxidized by solutions of other metal salts;
This reaction may be observed by placing copper wire in a solution containing a dissolved silver salt. Silver ions in solution are reduced to elemental silver at the surface of the copper wire, and the resulting Cu2+ ions dissolve in the solution to yield a characteristic blue color.

In the images above a copper wire is shown next to a solution containing silver(I) ions (a), and the displacement of dissolved silver ions by copper ions results (b), and an accumulation of gray-colored silver metal on the wire and the development of blue color in the solution, is due to dissolved copper ions (c).
There are three main types of chemical reactions: precipitation, acid-base, and oxidation-reduction reactions. Precipitation reactions always have a solid as a product and usually have two reactants and two products, which is called a double replacement reaction. Acid-base are the easiest to identify because they are reactions where the reactants are an acid and a base.
Oxidation-reduction reactions have the largest variety of reactions and mostly involve the transfer of electrons. The two main categories of oxidation-reduction reactions are combustion reactions and single replacement reactions. Combustion reactions usually involve oxygen gas as a reactant and carbon dioxide and water as products. Single replacement reactions have two reactants and two products (usually) and an element is found both as a reactant and a product.
Source: THIS TUTORIAL HAS BEEN ADAPTED FROM OPENSTAX “CHEMISTRY: ATOMS FIRST 2E”. ACCESS FOR FREE AT Chemistry: Atoms First 2e. LICENSE: CREATIVE COMMONS ATTRIBUTION 4.0 INTERNATIONAL