Table of Contents |
We learned about how wrapping your hands around a warm cup of tea lets you feel the heat spreading through your fingers. This is a simple but powerful example of heat transfer, which can be explained as the movement of thermal energy from one place to another due to a difference in temperature.
Heat transfer can happen quickly, like when a cooking pan heats up on a stove, or slowly, like when a picnic cooler is kept cold inside. We can influence how fast or slow heat moves by making thoughtful choices, such as using insulating materials (e.g., thick wool in winter), reducing airflow with weather stripping around doors, and selecting colors that reflect or absorb heat (e.g., a white roof that helps keep a house cooler in summer). Heat transfer is so common in our daily lives that it’s hard to imagine a situation where it doesn’t occur at all. Yet, no matter the setting, every instance of heat transfer happens through just three fundamental methods: conduction, convection, and radiation.
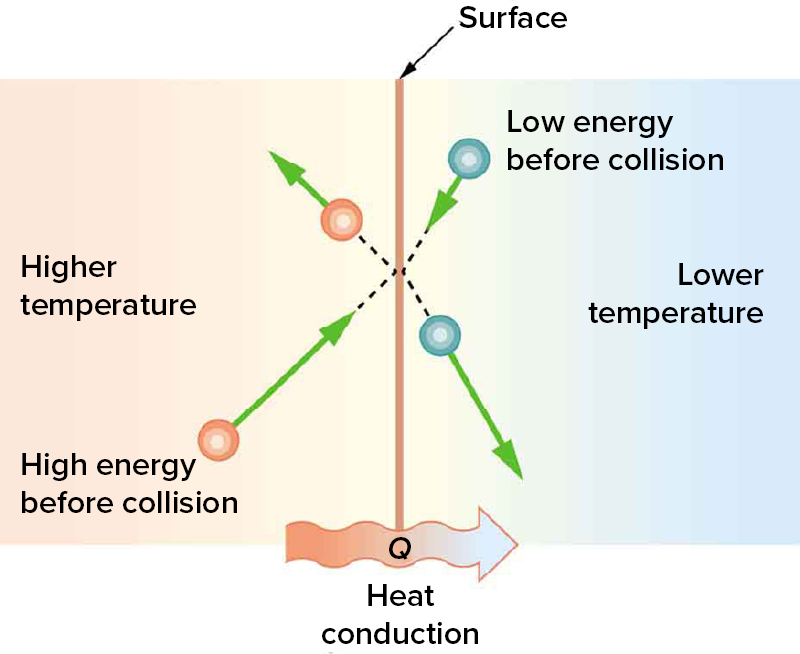
Conduction is the transfer of heat through direct contact. It happens when particles in a warmer object bump into particles in a cooler one, passing on their energy, like a gentle game of “pass the heat.” This process works best in solids, especially metals, because their particles are packed closely together and can transfer energy quickly.
Think about a time when you touched something warm or cold. What was it? Maybe a heated seat, a cold metal railing, or a laptop that got warm after hours of use. In each case, heat moved from one object to another through conduction. Heat conduction is influenced by both the properties of the material and several other key factors, including:
1. Thermal conductivity – How easily a material allows heat to pass through it.
Imagine sitting on a wooden bench and then moving to a metal one on a chilly day; both benches may be the same temperature, yet the metal feels much colder. Why is that? The answer lies in how different materials transfer heat. Metal draws heat away from your body much faster than wood does, making your skin feel colder even though the temperatures are the same. This difference in sensation is due to the rate of heat transfer.
Some materials allow heat to pass through them much more easily than others. For example, metals like copper, aluminum, and silver are excellent at transferring heat, while materials like wood, plastic, and rubber are not. As depicted in the diagram below, at a microscopic level, heat is transferred when faster-moving particles in a warmer object collide with slower-moving particles in a cooler one, passing on some of their energy.
2. Cross-sectional area – A larger contact area allows more heat to flow.
The size of the contact area also matters: placing your whole hand on a cold surface will cool it down faster than just touching it with a fingertip because more particles are interacting and transferring energy at once.
3. Temperature difference – Greater differences lead to faster heat transfer.
The greater the temperature difference between two objects, the faster this energy transfer happens. That’s why touching boiling water can cause a more severe burn than touching hot tap water. They are both hot, but boiling water has more thermal energy to transfer.
4. Thickness of the material – Thicker materials slow down the rate of conduction.
The thickness of a material plays a significant role in how quickly heat is conducted through it. Thicker materials act as better barriers to heat flow because thermal energy has to travel a longer distance to pass through. For example, a thick wool blanket keeps you warmer than a thin one because it slows down the escape of body heat to the cooler surroundings.
5. Duration of contact – The longer two objects are in contact, the more heat is transferred.
The amount of time two objects remain in contact affects how much heat is transferred between them. The longer the contact, the more energy has time to move from the warmer object to the cooler one. For instance, if you hold a warm mug for just a second, your hand barely warms up. However, if you hold it for a minute, you will feel the heat more clearly.
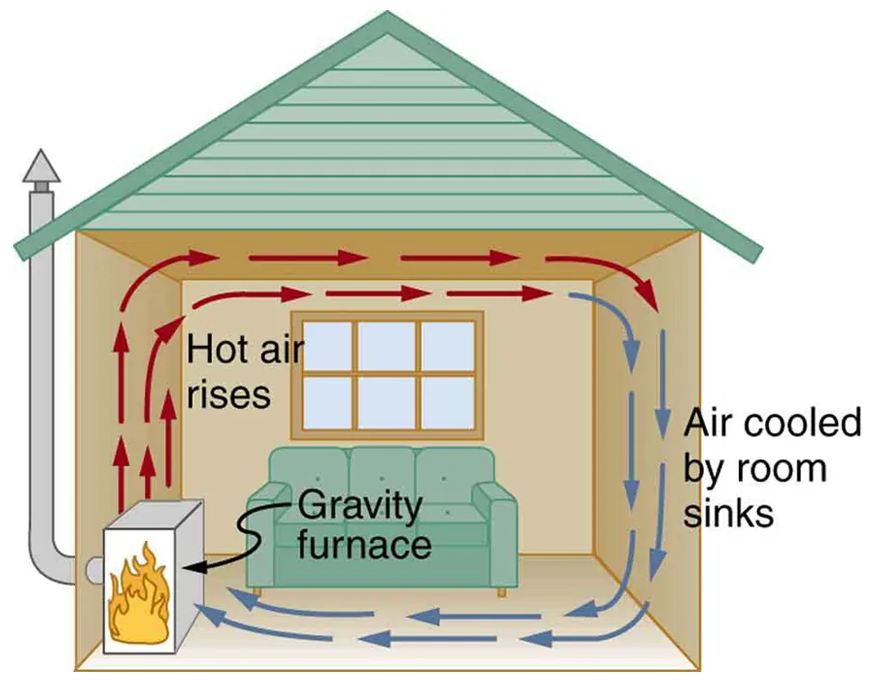
Convection is a method of heat transfer that occurs in fluids, liquids, and gases through the movement of particles. When a fluid is heated, the warmer, less dense regions rise while cooler, denser regions sink, creating a continuous circulation pattern. This process helps distribute heat more evenly. You can observe convection in action when watching water boil: bubbles rise from the bottom as hot water moves upward, while cooler water flows down to take its place. Convection is also responsible for natural phenomena like ocean currents, wind patterns, and even the way heat spreads through the air in a heated room.
Radiation is a way heat can travel without needing any contact or even air. It works by sending energy directly from a warm object to a cooler one. For example, the Sun warms your skin even though it’s millions of kilometers away. Unlike conduction or convection, radiation doesn’t need particles to carry the heat. It can move through empty space. This makes it especially important in situations like space travel, heating systems, and even how buildings stay warm.

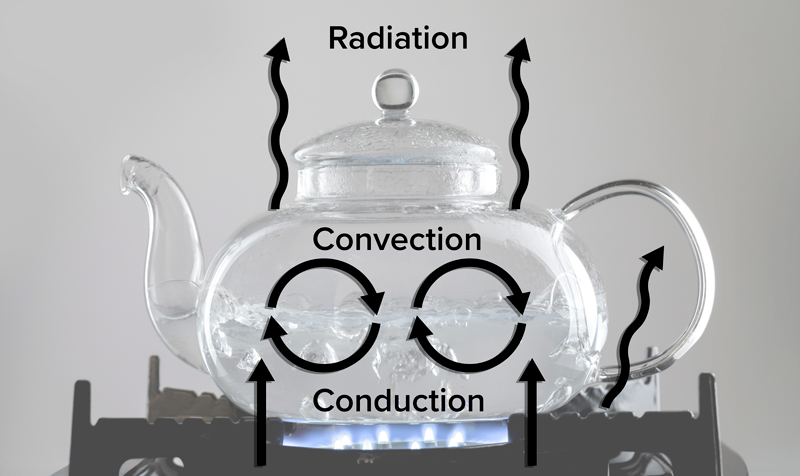
When water boils inside a kettle, all three methods of heat transfer are at work. Conduction happens first as heat moves from the stove into the metal base of the kettle. The metal conducts heat efficiently, warming the water inside. Then, convection takes over within the water. Hot water near the bottom rises while cooler water sinks, creating a circulation loop that evenly distributes heat. Finally, radiation occurs as heat is emitted from the hot surface of the kettle into the surrounding air. This combination of conduction, convection, and radiation allows the water to boil efficiently and the kettle to warm up the space around it.
SOURCE: THIS TUTORIAL HAS BEEN ADAPTED FROM (1) OPENSTAX “PHYSICS HIGH SCHOOL.” ACCESS FOR FREE AT OPENSTAX.ORG/DETAILS/BOOKS/PHYSICS/. (2) OPENSTAX “COLLEGE PHYSICS 2E.” ACCESS FOR FREE AT OPENSTAX.ORG/DETAILS/BOOKS/COLLEGE-PHYSICS-2E. (3) OPENSTAX “CHEMISTRY 2E”. ACCESS FOR FREE AT OPENSTAX.ORG/DETAILS/BOOKS/CHEMISTRY-2E (4) OPENSTAX “CHEMISTRY ATOMS FIRST 2E”. ACCESS FOR FREE AT OPENSTAX.ORG/DETAILS/BOOKS/CHEMISTRY-ATOMS-FIRST-2E LICENSING (1, 2, 3 & 4) CREATIVE COMMONS ATTRIBUTION 4.0 INTERNATIONAL.