Table of Contents |
It is often the case that a quantity of interest may not be easy (or even possible) to measure directly but instead must be calculated from other directly measured properties and appropriate mathematical relationships. This is a versatile mathematical approach known as dimensional analysis.
A ratio of two equivalent quantities expressed with different measurement units can be used as a unit conversion factor. For example, the lengths of 2.54 cm and 1 in. are equivalent, and so a unit conversion factor may be derived from the ratio,
Several other commonly used conversion factors are given in the table below.
| Length | Volume | Mass |
|---|---|---|
| 1 m = 1.0936 yd | 1 L = 1.0567 qt | 1 kg = 2.2046 lb |
| 1 in. = 2.54 cm (exact) | 1 qt = 0.94635 L | 1 lb = 453.59 g |
| 1 km = 0.62137 mi |
1 ft = 28.317 L = 28.317 L
|
1 (avoirdupois) oz = 28.349 g |
| 1 mi = 1609.3 m | 1 tbsp = 14.787 mL | 1 (troy) oz = 31.103 g |
When a quantity (such as distance in inches) is multiplied by an appropriate unit conversion factor, the quantity is converted to an equivalent value with different units (such as distance in centimeters).
EXAMPLE
A basketball player’s vertical jump of 34 inches can be converted to centimeters as follows:Beyond simple unit conversions, dimensional analysis can be used to solve more complex problems involving computations. Regardless of the details, the basic approach is the same—all the factors involved in the calculation must be appropriately oriented to ensure that their labels (units) will appropriately cancel and/or combine to yield the desired unit in the result. As your study of chemistry continues, you will encounter many opportunities to apply this approach.
EXAMPLE
If 4.00 qts of antifreeze weighs 9.26 lbs, what is the density in kilograms/L?Temperature is an intensive property. An intensive property is a property that does not depend on the quantity of matter. We use the word temperature to refer to the hotness or coldness of a substance.
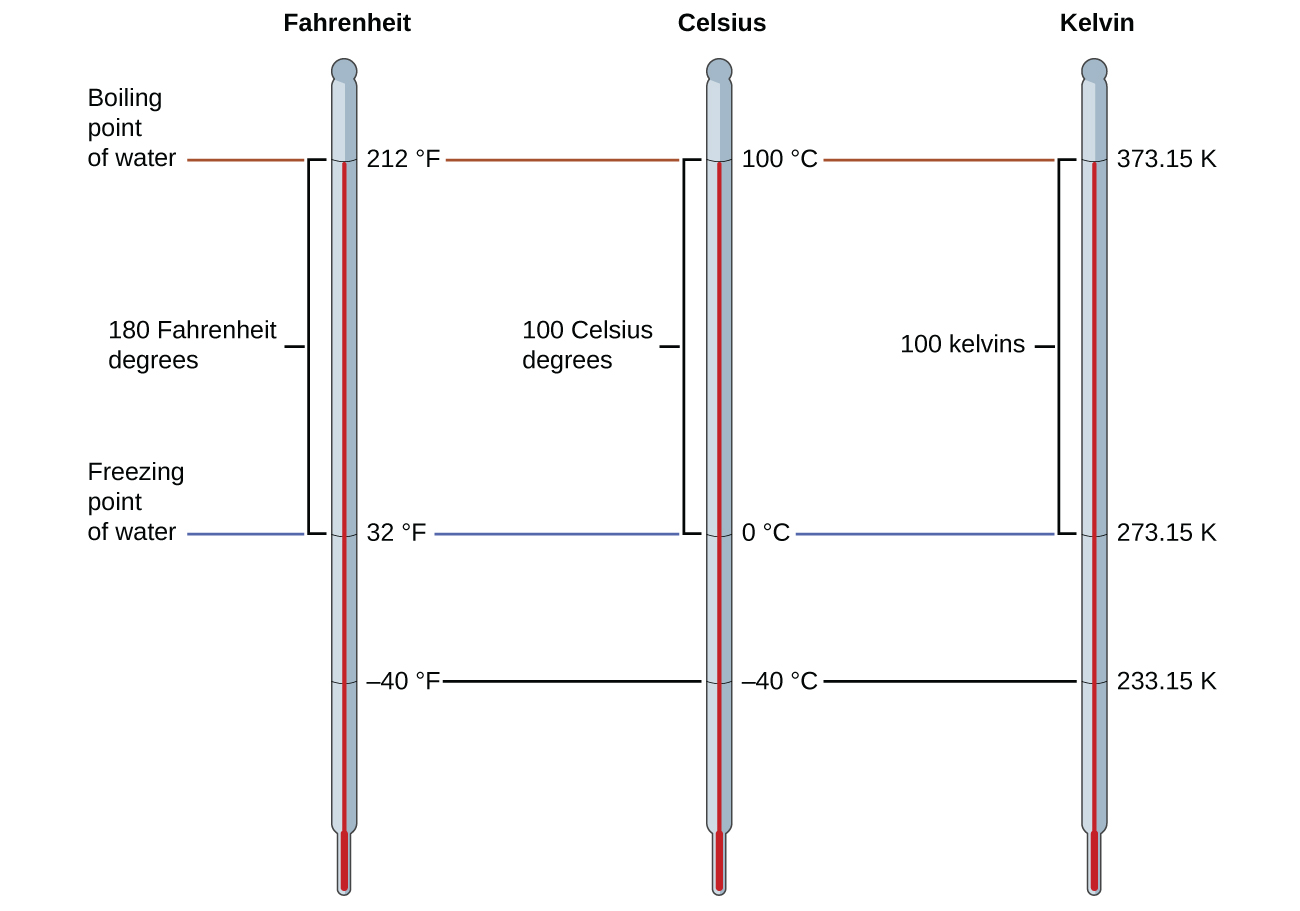
Temperature scales are defined relative to selected reference temperatures. Two of the most commonly used are the freezing and boiling temperatures of water at a specified atmospheric pressure. On the Celsius scale, 0 °C is defined as the freezing temperature of water and 100 °C as the boiling temperature of water. On the Fahrenheit scale, the freezing point of water is defined as 32 °F and the boiling temperature as 212 °F.
The relationship between the Celsius and Fahrenheit temperature does not have a simple conversion like some of the earlier examples, such as 1 in. = 2.54 cm. The relationship between these temperature scales can be written as a formula as seen below.
Unlike the Celsius and Fahrenheit scales, the Kelvin scale is an absolute temperature scale in which 0 (zero) K corresponds to the lowest temperature that can theoretically be achieved. Since the Kelvin temperature scale is absolute, a degree symbol is not included in the unit abbreviation, K. There is a constant value (273.15) between kelvin and Celsius, which gives the following relationship.
Source: THIS TUTORIAL HAS BEEN ADAPTED FROM OPENSTAX “CHEMISTRY: ATOMS FIRST 2E”. ACCESS FOR FREE AT Chemistry: Atoms First 2e. LICENSE: CREATIVE COMMONS ATTRIBUTION 4.0 INTERNATIONAL