Table of Contents |
In nature, very few atoms are stable enough to exist alone. Instead, atoms stabilize through interactions between the electrons in their valence shells. These interactions form chemical bonds. A bond is an attraction that holds atoms in the same vicinity.
Three types of chemical bonds are important in human physiology, because they hold together substances that are used by the body for critical aspects of homeostasis, signaling, and energy production, to name just a few important processes. These are ionic bonds, covalent bonds, and hydrogen bonds.
In a previous lesson, you learned about the formation of ions by the donation (shedding) or acceptance of electrons to stabilize an atom. A donation, or giving away, of one or more electrons leads to the production of a positive ion or cation. Acceptance of one or more electrons leads to the production of a negative ion or anion.
Anions and cations, when in close proximity exert a strong attraction due to their opposite electrical charges. This forms an ionic bond, an ongoing, close association between ions of opposite charges. The resulting compound is referred to as a salt.
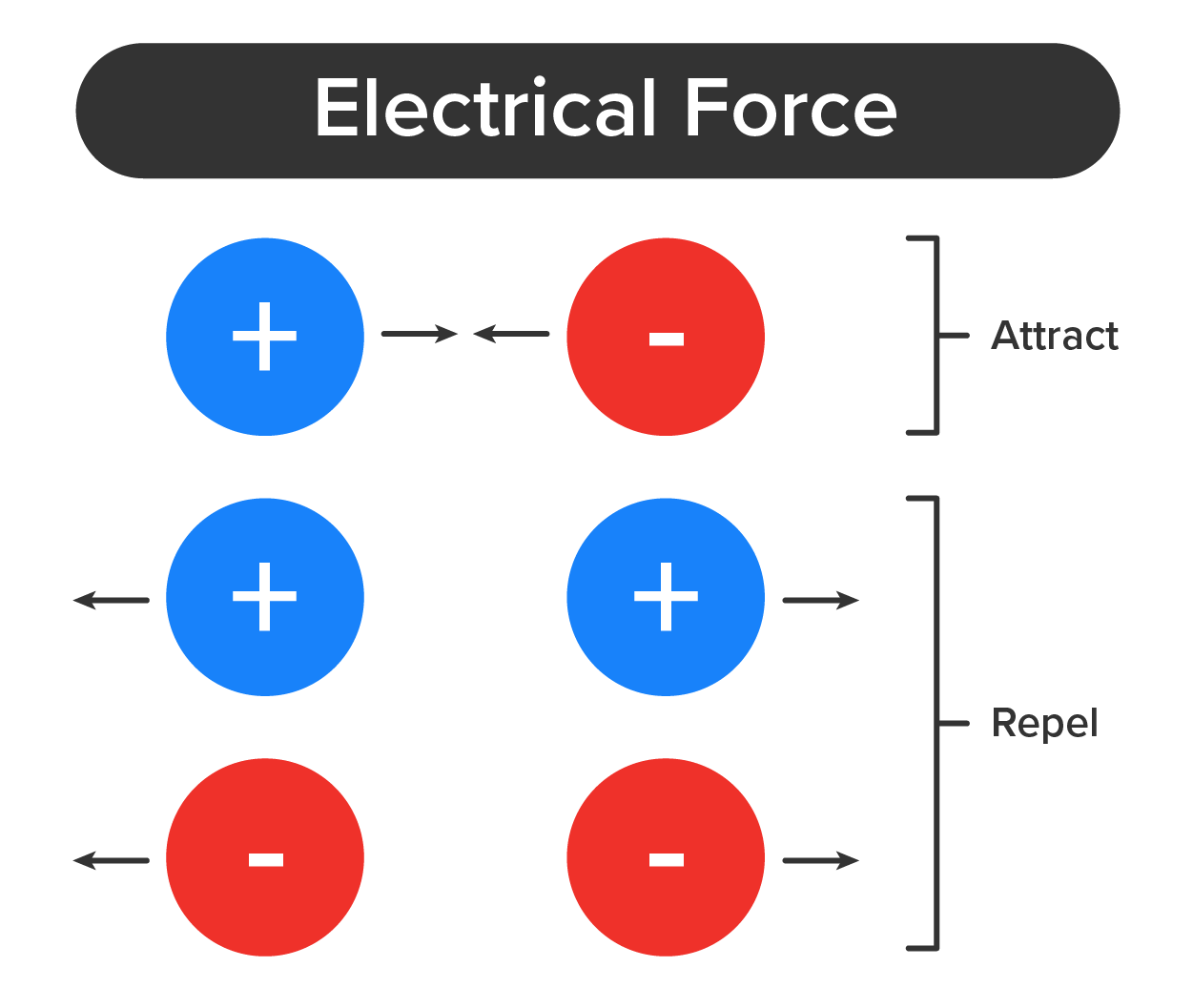
EXAMPLE
The table salt you sprinkle on your food, sodium chloride, owes its existence to ionic bonding. As shown in the image below, sodium commonly donates an electron to chlorine, becoming the sodium cation Na⁺>. When chlorine accepts the electron, it becomes the chloride anion, Cl⁻>. With their opposing charges, these two ions strongly attract each other.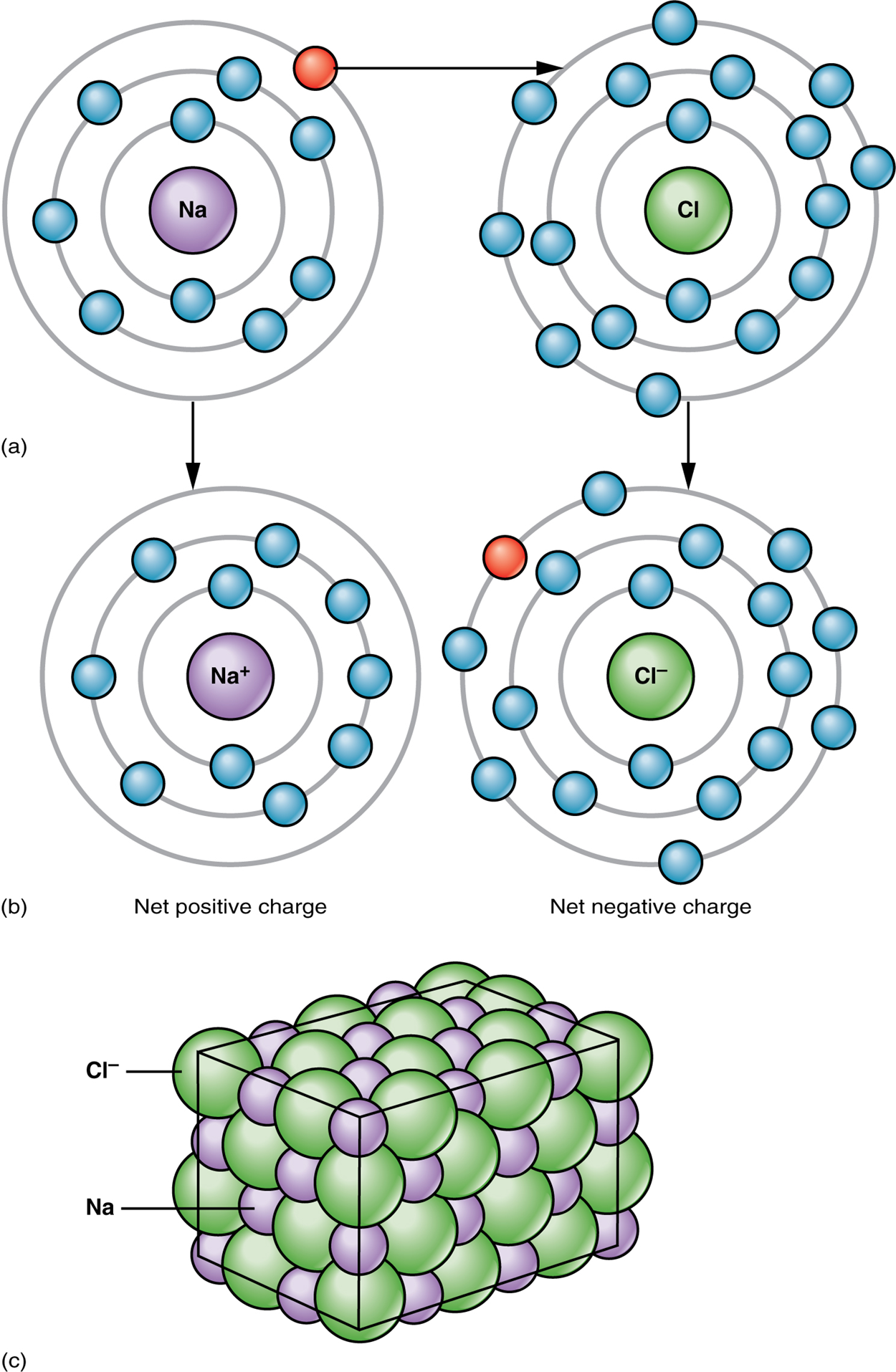
While ionic bonds are formed by atoms that have already stabilized themselves by altering their valence shell, some atoms are unable to do so and require bonding in order to stabilize. These atoms do not have enough energy to pull an electron off another atom and are too strong to have their electrons pulled off.
Bonded atomic structures formed by covalent bonds share electrons in a mutually stabilizing relationship.
EXAMPLE
Imagine that you moved into an apartment with a roommate. This person now lives in close proximity to you. When you moved in, you both had one oven mitt each. However, you both have been burnt in the past by using only one oven mitt to pull hot dishes out of the oven before. It is far safer (i.e., stable) to use two oven mitts at the same time.In the images below you see several common types of covalent bonds based on how many electrons are shared. In a single covalent bond, a single electron pair is shared between two atoms (one per atom), while in a double covalent bond, two pairs of electrons are shared between two atoms. There even are triple covalent bonds, where three electron pairs are shared between two atoms. In any of these cases, the electrons are shared to fill the valence shells of both atoms, ultimately stabilizing both of the atoms involved by completing the octet rule (or the first electron shell in the case of hydrogen gas (H₂).
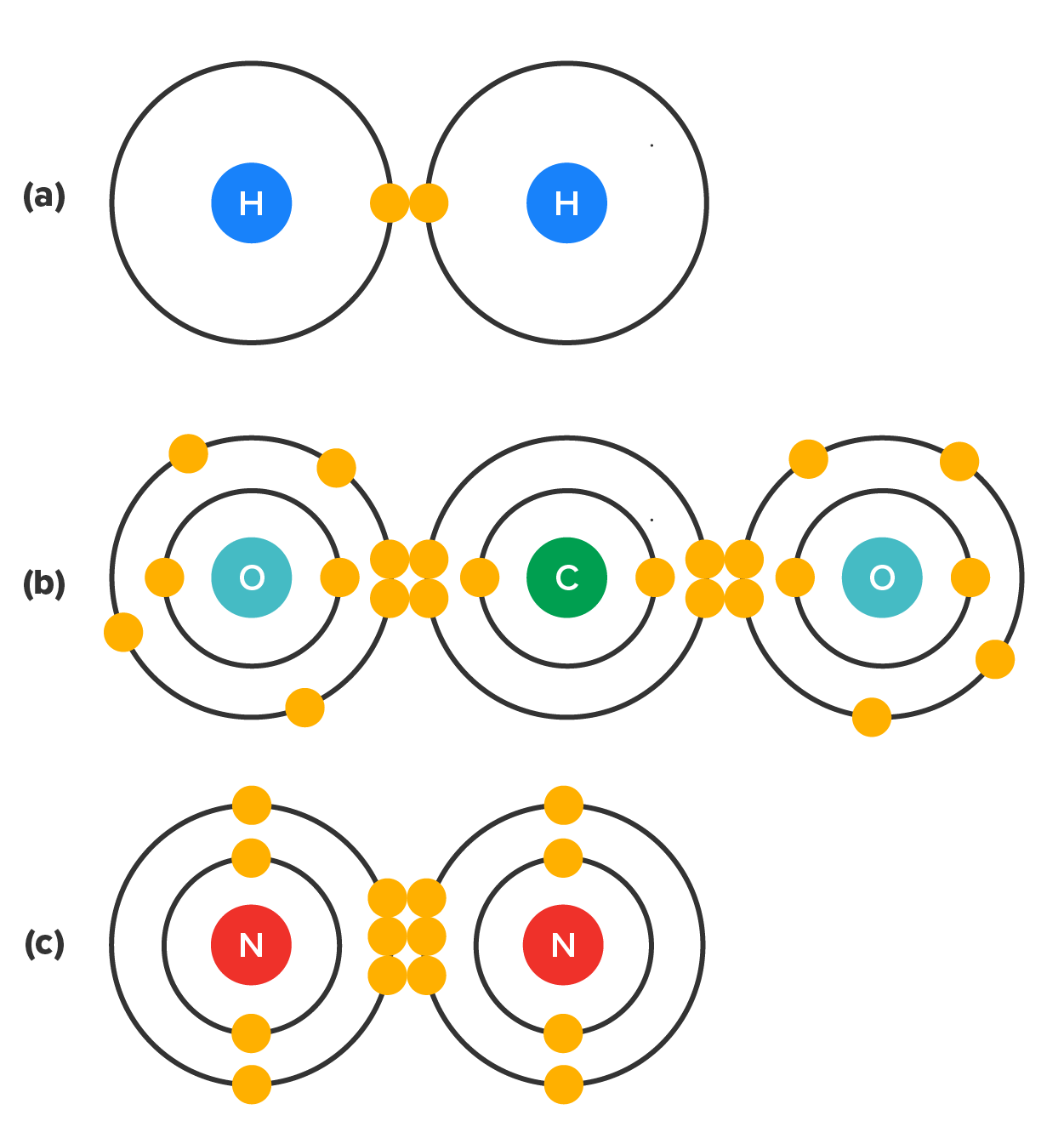
You can see that the covalent bonds shown in the image above are balanced. The number of positive and negative charges on either side of the bond are relatively equal, allowing the shared electrons to spend equal amounts of time around both atoms. Because of this, there are no poles or opposing ends and all charges, both positive and negative are equal. This type of covalent bond which shares electrons equally between two atoms is called a nonpolar covalent bond.
Groups of legislators with completely opposite views on a particular issue are often described as “polarized” by news writers. Opposite ends of a magnet, called poles, have different characteristics from one another. In chemistry, a polar molecule is one that contains regions that have opposite electrical charges. Bonded atomic structures with polar covalent bonds share electrons unequally.
The most familiar example of a polar molecule is water (see image below). The molecule has three parts: one atom of oxygen, the nucleus of which contains eight protons, and two hydrogen atoms, whose nuclei each contain only one proton. Because every proton exerts an identical positive charge, the nucleus of oxygen exerts a force eight times greater than the nucleus of hydrogen. This means that the negatively charged electrons present in the water molecule are more strongly attracted to the oxygen nucleus than to the hydrogen nuclei. The electrons, therefore, spend more time “spinning” around the oxygen atom than the hydrogen atom. This causes the oxygen atom to be partially negative while each hydrogen is partially positive, setting up ends with opposing electrical charges; poles. As shown in the image below, regions of weak polarity are indicated with the Greek letter delta (δ) and a plus (+) or minus (–) sign.
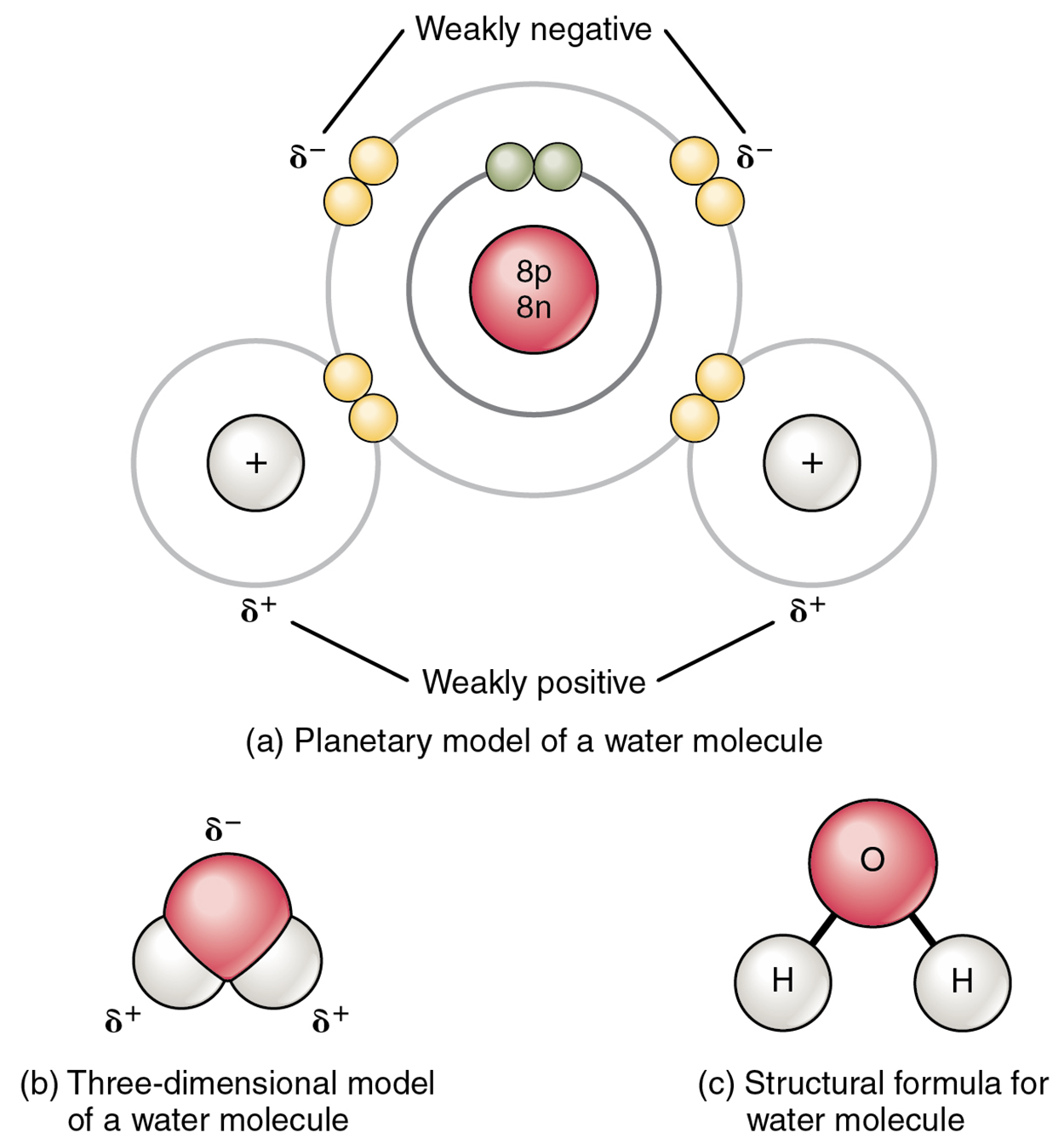
A hydrogen bond is formed when a partially positive hydrogen atom in a polar molecule already bonded to one electronegative atom (i.e., oxygen or nitrogen) is attracted to another electronegative atom from another molecule. Because both atoms are only partially charged, their attraction, and therefore hydrogen bonds, are weak individually.
The most common example of hydrogen bonding in the natural world occurs between molecules of water. It happens before your eyes whenever two raindrops merge into a larger bead, or a creek spills into a river. Hydrogen bonding occurs because the weakly negative oxygen atom in one water molecule is attracted to the weakly positive hydrogen atoms of two other water molecules (see the image below).
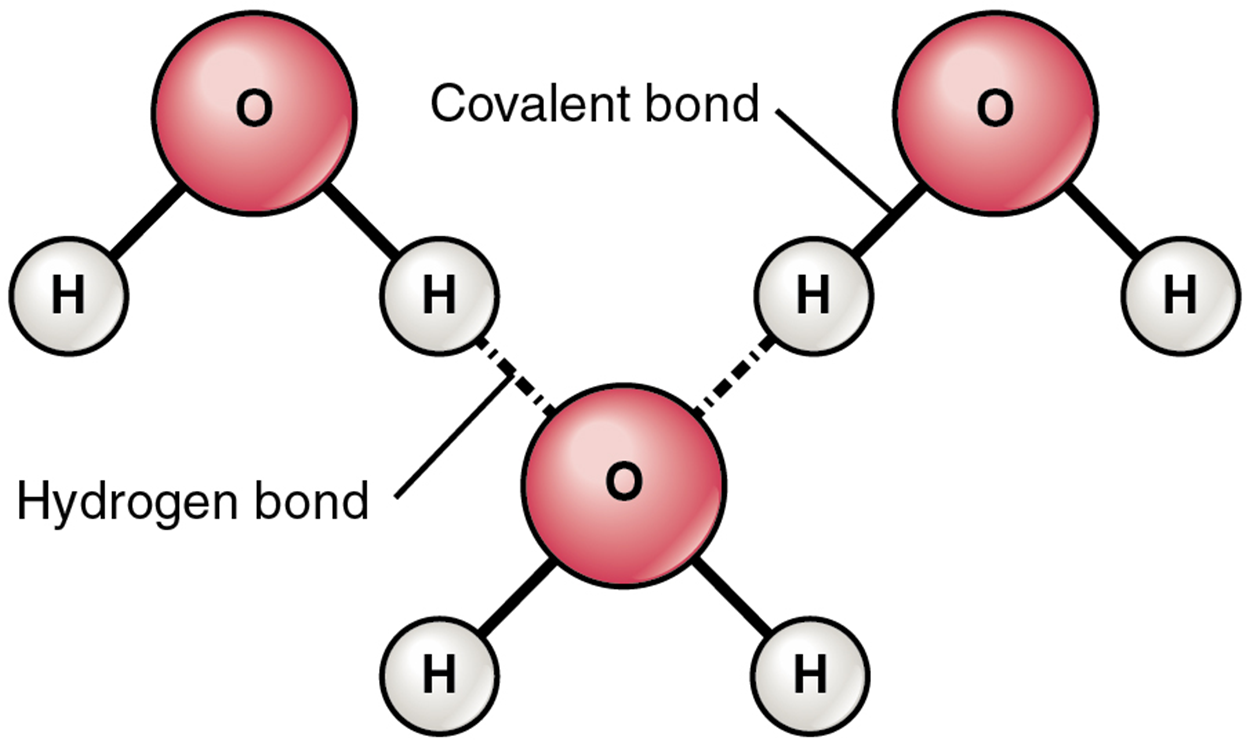
Water molecules also strongly attract other types of charged molecules such as ions. This explains why “table salt” (sodium chloride), for example, actually is called a “salt” in chemistry, which consists of equal numbers of positively-charged sodium (Na⁺) and negatively-charged chloride (Cl⁻), dissolves so readily in water. The dipole water molecule is attracted to the dipole ends of sodium chloride. These interactions affect the ionic bond and dissociate it (break it apart).
Water molecules also repel other molecules with nonpolar covalent bonds, like fats, lipids, and oils. You can demonstrate this with a simple kitchen experiment: pour a teaspoon of vegetable oil, which is formed by nonpolar covalent bonds, into a glass of water. Instead of instantly dissolving in the water, the oil forms a distinct bead because the polar water molecules repel the nonpolar oil.
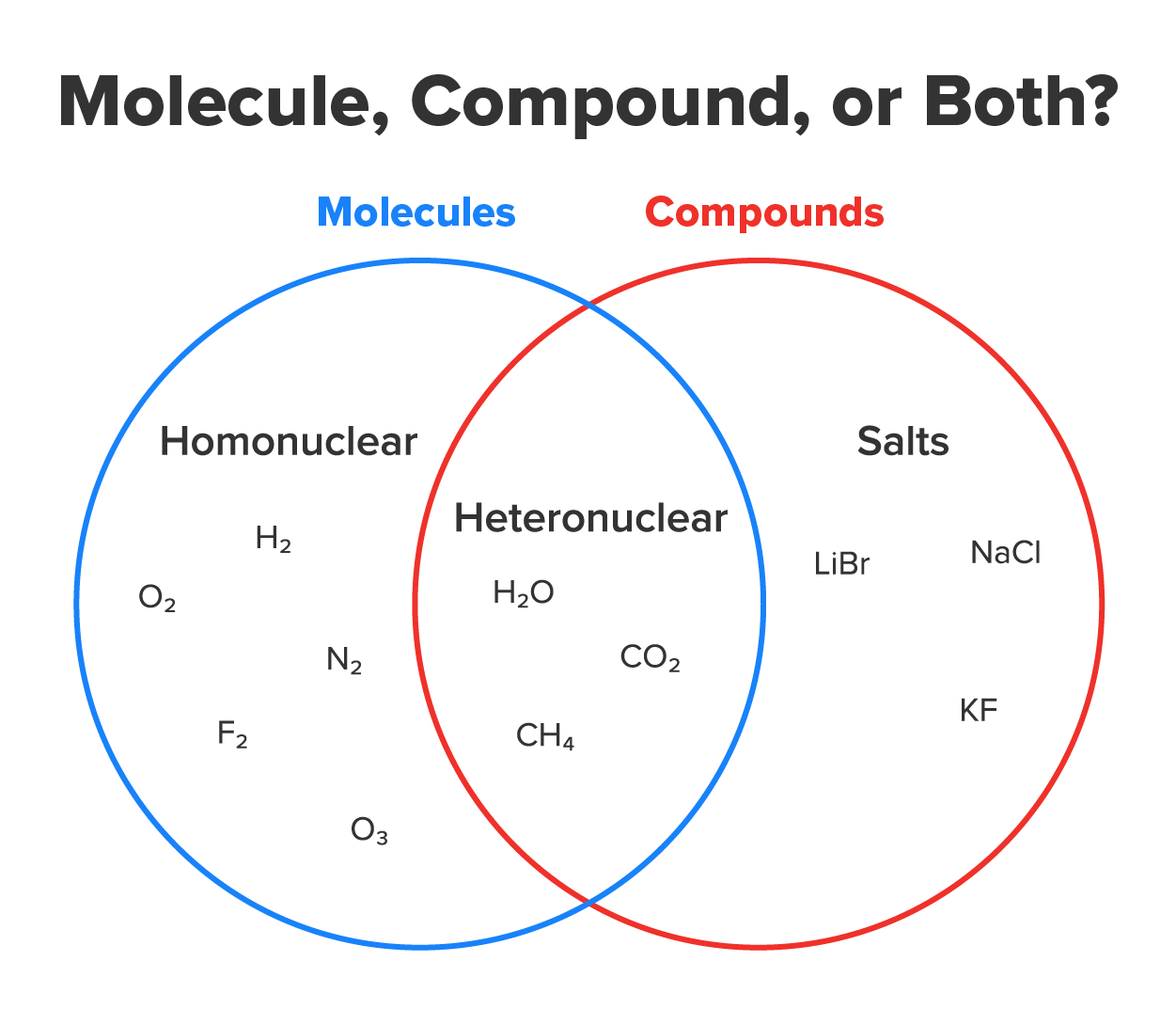
When atoms react to form the bonds described above, they form groupings that are more stable than their component atoms. These groupings are commonly called molecules and/or compounds.
Molecules are groups of two or more atoms connected by a covalent bond. The oxygen you breathe (O₂), the carbon dioxide (CO₂) you produce, and water (H₂O) you drink are all molecules. Molecules can be further categorized based on what types atoms they include—all the same or different. Molecules that contain atoms of only one element such as H₂, O₂, or N₂ are homonuclear molecules (homo, the same). Molecules that contain atoms of two or more elements such as H₂O, CO₂, or CH₄ are heteronuclear molecules (hetero, different).
Compounds are groups of two or more atoms of different elements joined by any type of bond in a fixed proportion. All heteronuclear molecules (H₂O, CO₂, CH₄, and others) are also compounds. However, homonuclear molecules (H₂, O₂, N₂, and others) are not compounds because they only contain atoms from one type of element. Salts such as sodium chloride (NaCl, table salt) are formed by ionic bonds and are therefore classified as compounds but not molecules.
Source: THIS TUTORIAL HAS BEEN ADAPTED FROM OPENSTAX “ANATOMY AND PHYSIOLOGY 2E.” ACCESS FOR FREE AT HTTPS://OPENSTAX.ORG/DETAILS/BOOKS/ANATOMY-AND-PHYSIOLOGY-2E. LICENSE: CC ATTRIBUTION 4.0 INTERNATIONAL.