Table of Contents |
Carbohydrates are organic molecules composed of carbon, hydrogen, and oxygen atoms. The family of carbohydrates includes both simple and complex sugars.
EXAMPLE
Glucose and fructose are examples of simple sugars, and starch, glycogen, and cellulose are all examples of complex sugars.The complex sugars are also called polysaccharides and are made of multiple monosaccharide molecules. Polysaccharides serve as energy storage (e.g., starch and glycogen) and as structural components (e.g., chitin in insect exoskeletons and cellulose in plant cell walls).
During digestion, carbohydrates are broken down into simple, soluble sugars that can be transported across the intestinal wall into the circulatory system to be transported throughout the body. Carbohydrate digestion begins in the mouth with the action of salivary amylase on starches and ends with monosaccharides being absorbed across the epithelium of the small intestine. Once the absorbed monosaccharides are transported to the tissues, the process of cellular respiration begins, during which ATP is produced from breaking down glucose. This lesson will focus first on glycolysis, a process where the monosaccharide glucose is oxidized, releasing the energy stored in its bonds to produce ATP.
Glucose is the body’s most readily available source of energy. After digestive processes break polysaccharides down into monosaccharides, including glucose, the monosaccharides are transported across the wall of the small intestine and into the circulatory system, which transports them to the liver. In the liver, hepatocytes either pass the glucose on through the circulatory system or store excess glucose as glycogen. Cells in the body take up the circulating glucose in response to insulin and, through a series of reactions called glycolysis, transfer some of the energy in glucose to ADP to form ATP. The last step in glycolysis produces the product pyruvate.
Now, the process of glycolysis begins. Glycolysis consists of 10 steps, but has three major phases that are most important to remember so that you grasp what is happening:
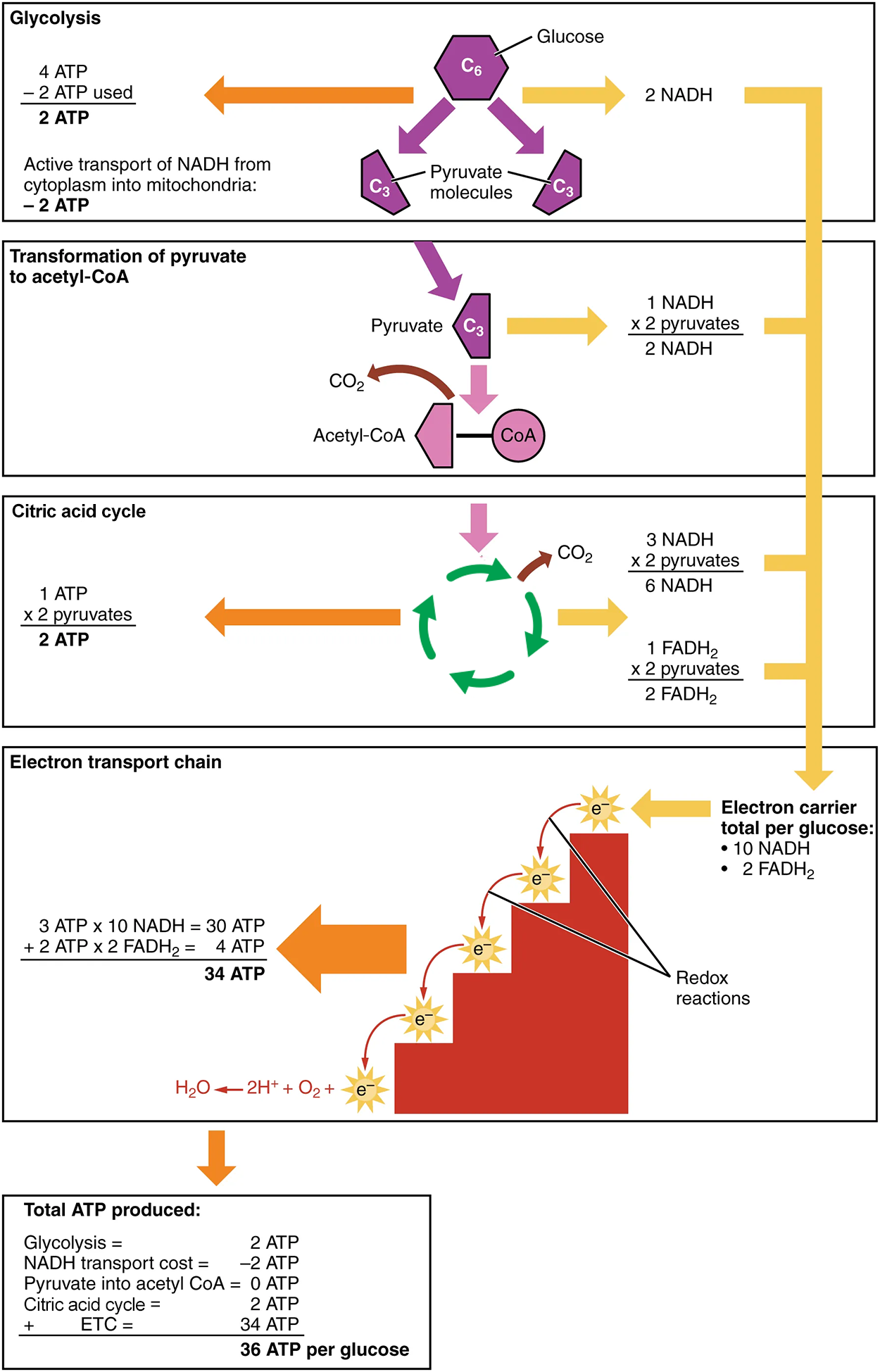
As shown in the image below, the net effect of glycolysis is that two ATP (four ATP from step 3 [rearrangement] and two ATP from step 1 [glycolysis] = two ATP), three NADH, and two pyruvate molecules are produced.
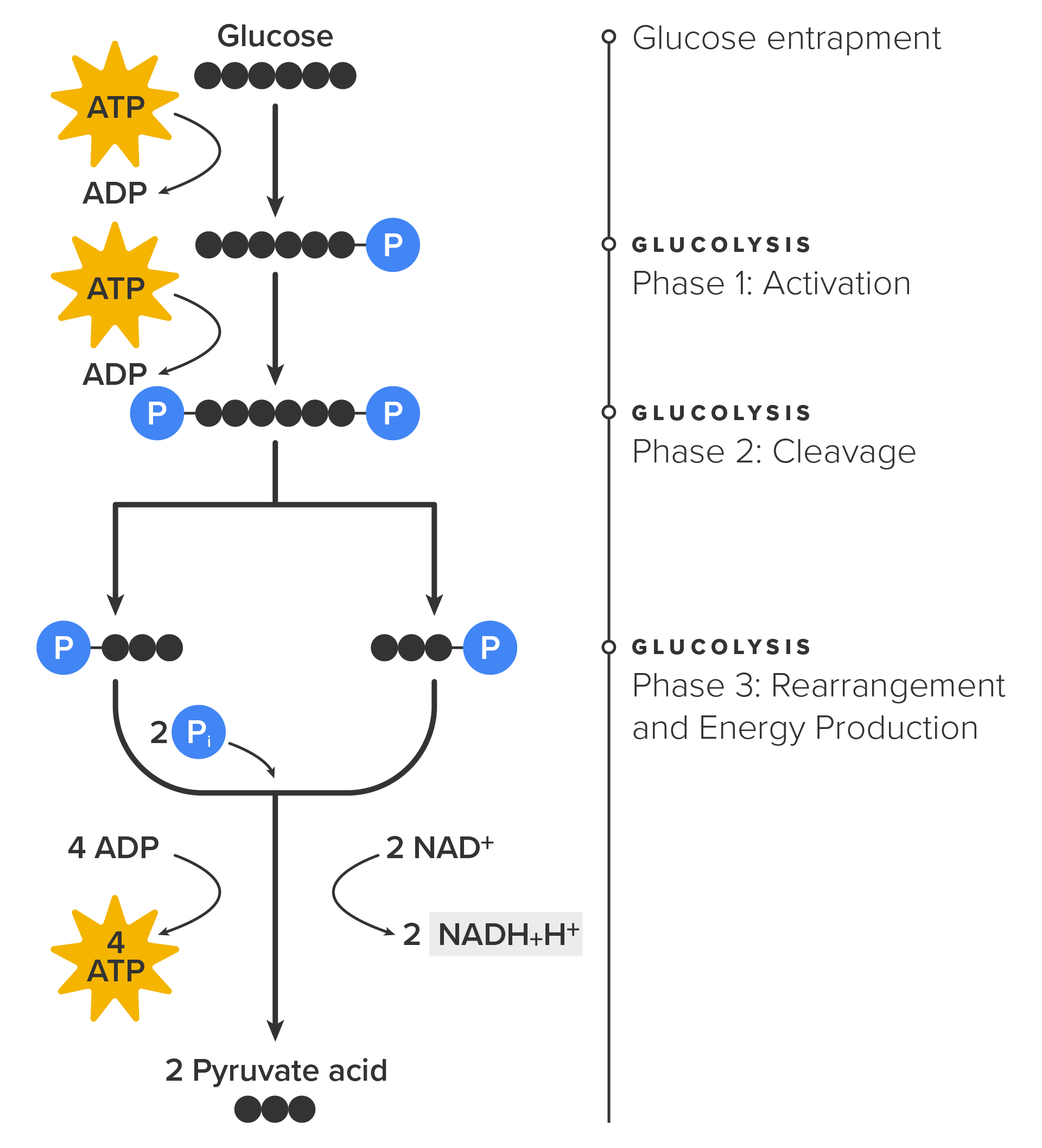
In summary, one glucose molecule breaks down into two pyruvate molecules and creates two net ATP molecules and two NADH molecules by glycolysis. Therefore, glycolysis generates energy for the cell and creates pyruvate molecules that can be processed further through the aerobic citric acid cycle (also called the Krebs cycle or tricarboxylic acid cycle/TCA cycle); converted into lactic acid or alcohol (in yeast) by fermentation (which is anaerobic); or used later to produce glucose through gluconeogenesis.
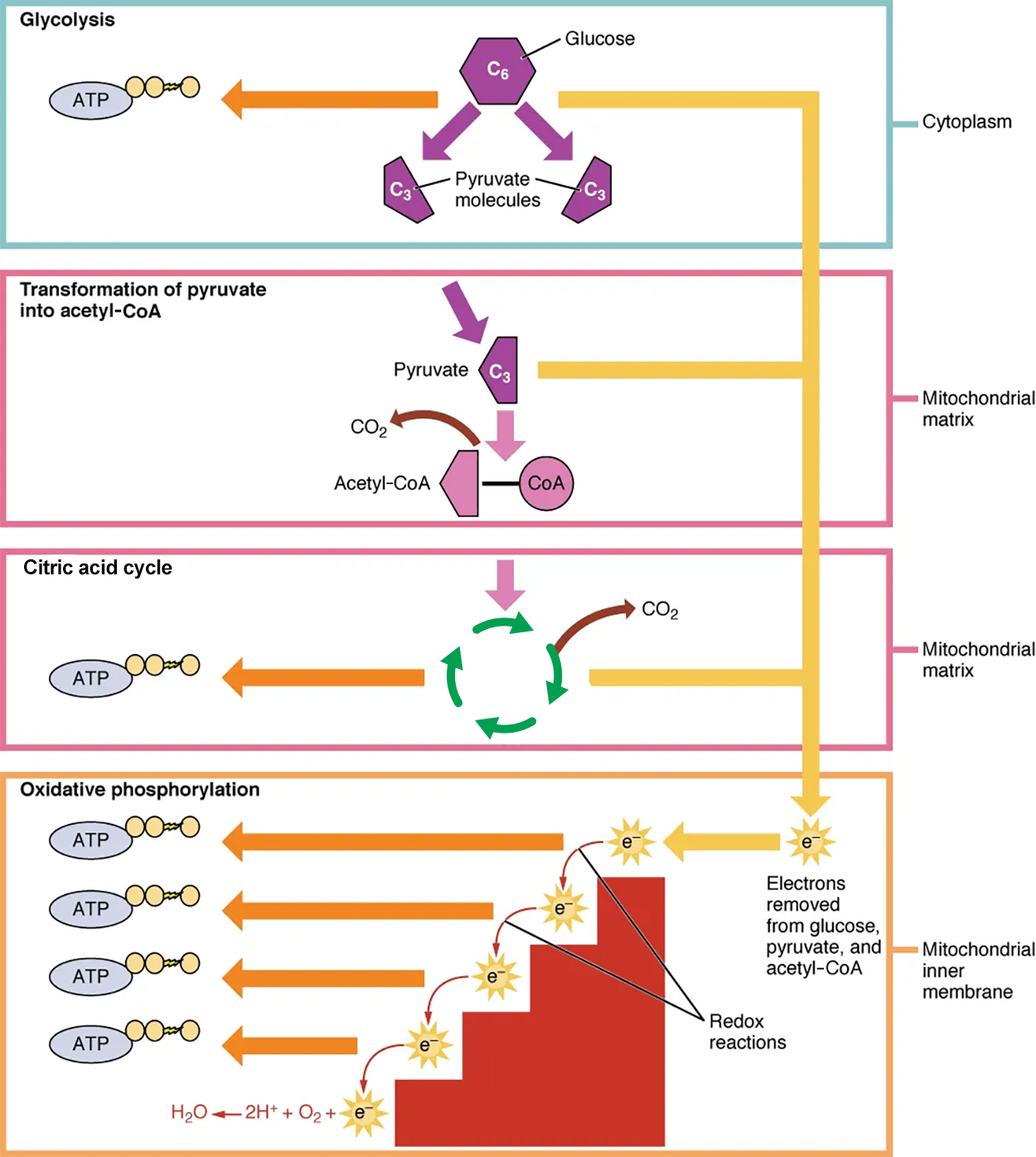
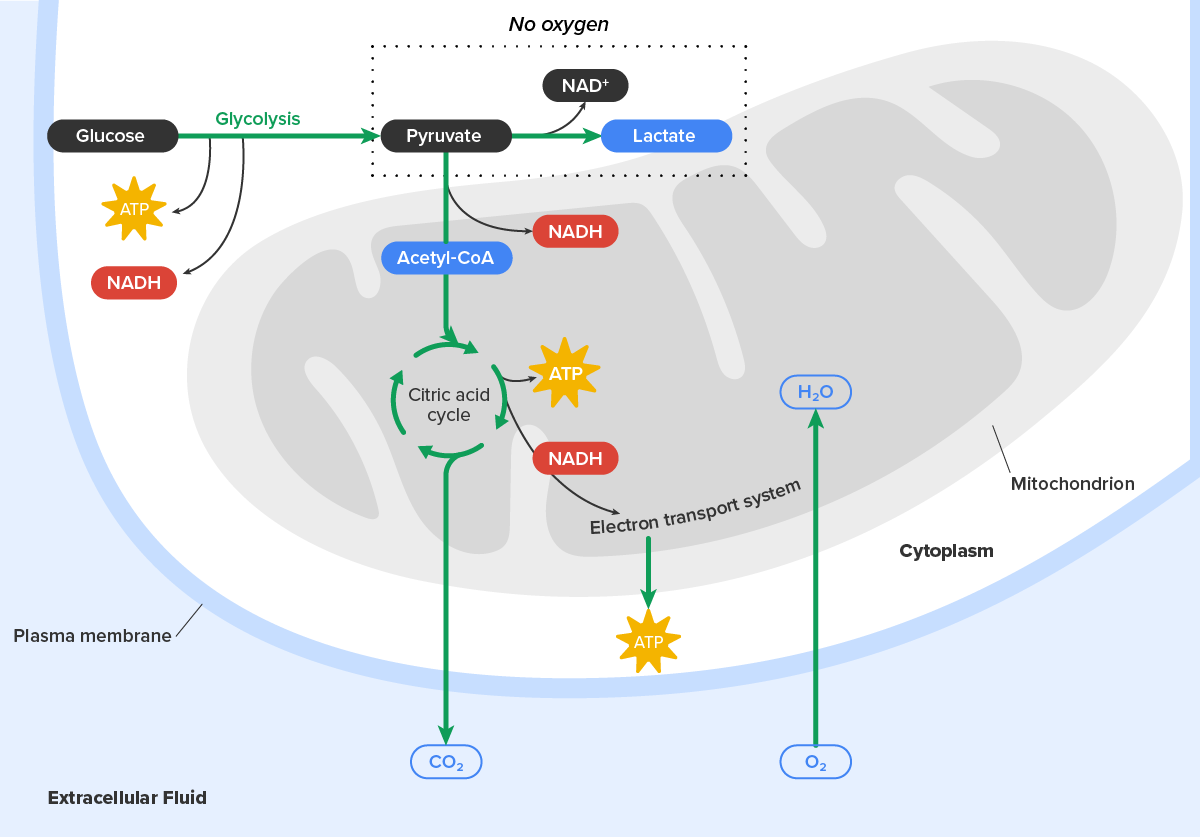
In the presence of oxygen, a carbon is removed from pyruvate, and a coenzyme is added to make acetyl coenzyme A (acetyl-CoA), which enters the citric acid cycle. The carbon that is removed is released as carbon dioxide. In the citric acid cycle, additional energy is extracted from acetyl-CoA as electrons are transferred to the receptors NAD⁺, GDP, and FAD, and more carbon dioxide is produced as a “waste product.” The NADH and FADH₂ pass electrons on to the electron transport chain, which uses the transferred energy to produce ATP; this process is where most of the energy from glucose catabolism is extracted. As the terminal step in the electron transport chain, oxygen is the terminal electron acceptor and creates water inside the mitochondria. By this time, all of the carbons from the original glucose have been released as carbon dioxide, energy has been stored in ATP, and oxygen has been consumed.
When oxygen is limited or absent, pyruvate enters an anaerobic pathway called fermentation. In these reactions, pyruvate can be converted into lactic acid. In addition to generating an additional ATP, this pathway serves to keep the pyruvate concentration low so glycolysis continues, and it oxidizes NADH into the NAD⁺ needed by glycolysis. In this reaction, lactic acid replaces oxygen as the final electron acceptor. Anaerobic respiration occurs in most cells of the body when oxygen is limited or mitochondria are absent or nonfunctional. For example, because erythrocytes (red blood cells) lack mitochondria, they must produce their ATP from glycolysis alone.
This is an effective pathway of ATP production for short periods of time, ranging from seconds to a few minutes. The lactic acid produced diffuses into the plasma and is carried to the liver, where it is converted back into pyruvate or glucose via the Cori cycle (also known as the lactic acid cycle). Similarly, when a person exercises, muscles use ATP faster than oxygen can be delivered to them. They depend on glycolysis and lactic acid production for rapid ATP production.
The pyruvate molecules generated during glycolysis are transported across the mitochondrial membrane into the inner mitochondrial matrix, where they are metabolized by enzymes in a pathway called the citric acid cycle. As noted above, the citric acid cycle is also commonly called the Krebs cycle or the tricarboxylic acid (TCA) cycle. During the citric acid cycle, high-energy molecules, including ATP, NADH, and FADH₂, are created. NADH and FADH₂ then pass electrons through the electron transport chain in the mitochondria to generate more ATP molecules.
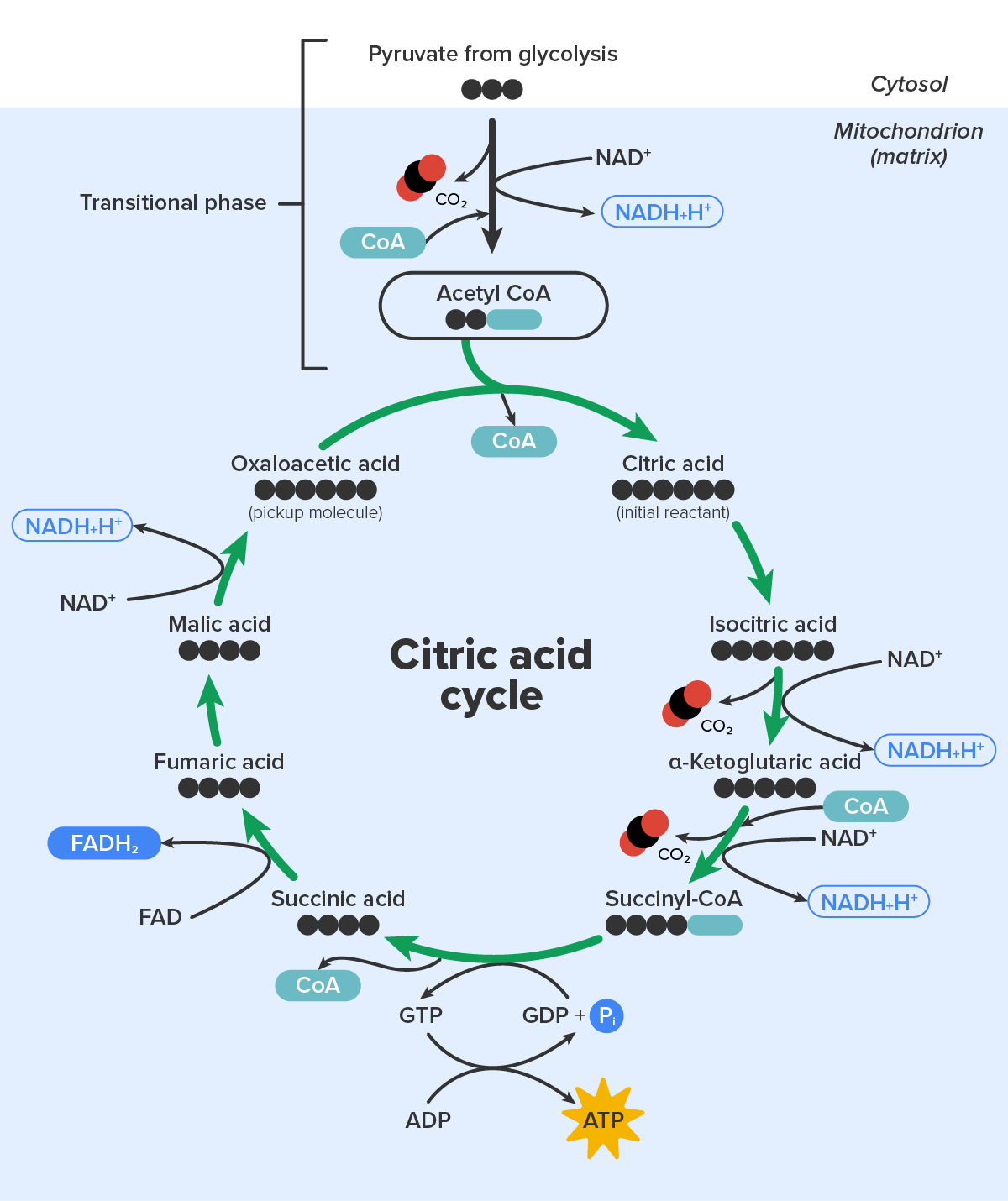
The three-carbon pyruvate molecule generated during glycolysis moves from the cytoplasm into the mitochondrial matrix, where it is converted by the enzyme pyruvate dehydrogenase into a two-carbon acetyl coenzyme A (acetyl-CoA) molecule.
The electron transport chain (ETC) uses the NADH and FADH₂ produced by glycolysis and the citric acid cycle to generate ATP. Electrons from NADH and FADH₂ are transferred through protein complexes embedded in the inner mitochondrial membrane by a series of enzymatic reactions.
The ETC consists of a series of four enzyme complexes (Complex I–Complex IV) and two coenzymes (ubiquinone and cytochrome c), which act as electron carriers and proton pumps used to transfer H⁺ from inside the inner membrane into the space between the inner and outer mitochondrial membranes. The ETC couples the transfer of electrons between a donor (like NADH) and an electron acceptor (like O₂) with the transfer of protons (H⁺ ions) across the inner mitochondrial membrane, enabling oxidative phosphorylation, which is the process that converts high-energy NADH and FADH₂ into ATP.
In the presence of oxygen, energy is passed, stepwise, through the electron carriers to gradually collect the energy needed to attach a phosphate to ADP and produce ATP. The role of molecular oxygen, O₂, is as the terminal electron acceptor for the ETC. This means that once the electrons have passed through the entire ETC, they must be passed to another, separate molecule. These electrons, O₂, and H⁺ ions from the matrix combine to form new water molecules.
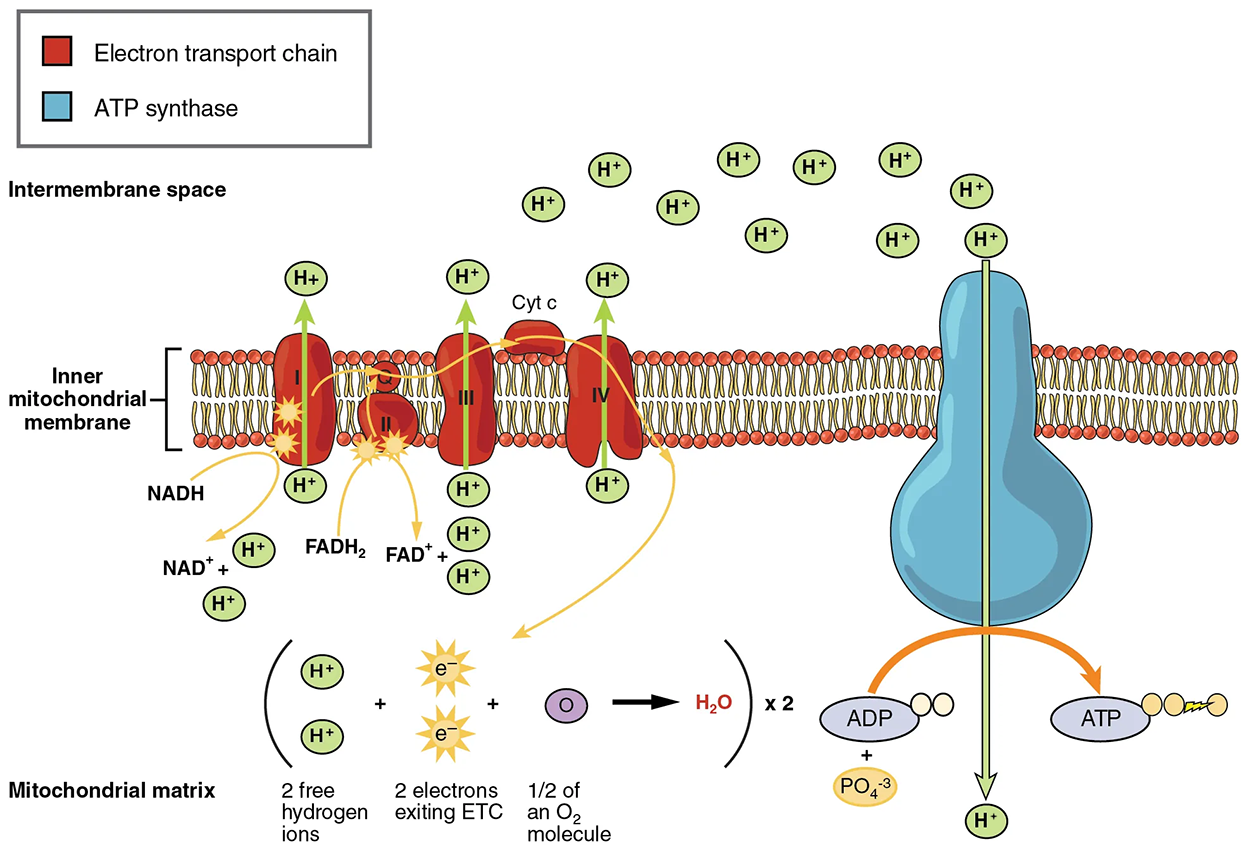
The electrons released from NADH and FADH₂ are passed along the chain by each of the carriers. Each of these reactions releases a small amount of energy, which is used to pump H⁺ ions across the inner membrane. Ultimately, the electrons are passed from the final carrier, cytochrome c, to molecular oxygen, which gets split and combined with protons in the matrix to make water. The accumulation of these protons in the space between the membranes creates a proton gradient with respect to the mitochondrial matrix in which the energy is stored.
Also embedded in the inner mitochondrial membrane is a protein pore complex called ATP synthase. ATP is made by this enzyme through the process of chemiosmosis, which couples the synthesis of ATP to the proton gradient across the membrane. Effectively, the ATP synthase is a turbine that is powered by the flow of H⁺ ions across the inner membrane down a gradient and into the mitochondrial matrix. As the H⁺ ions traverse the complex, the shaft of the complex rotates. This rotation enables other portions of ATP synthase to use the energy from the proton gradient to create ATP from ADP and Pᵢ. In accounting for the total number of ATP produced per glucose molecule through aerobic respiration, it is important to remember the following points:
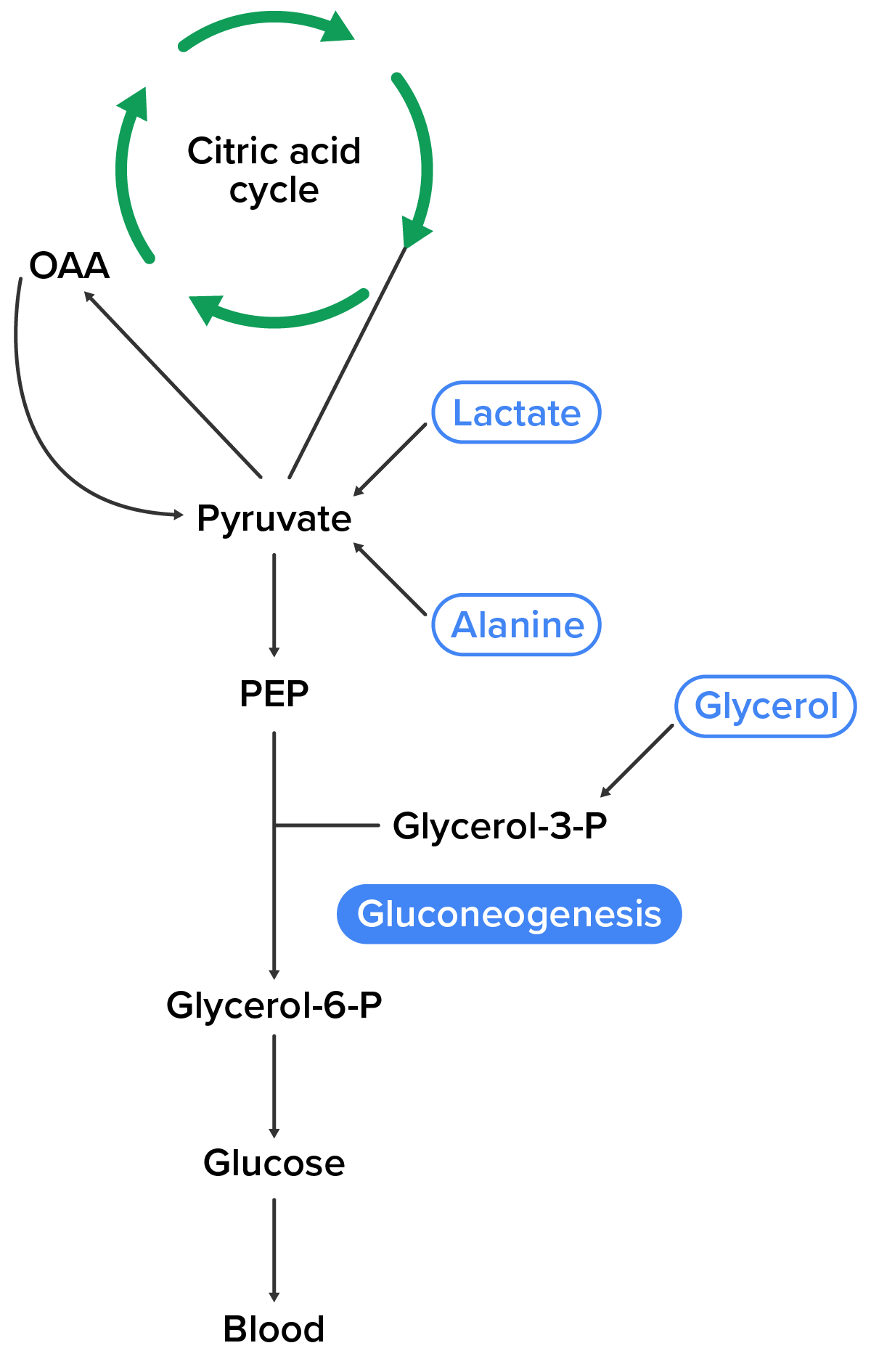
Gluconeogenesis is the synthesis of new glucose molecules from pyruvate, lactate, glycerol, or the amino acids alanine or glutamine. This process takes place primarily in the liver during periods of low glucose—that is, under conditions of fasting, starvation, and low carbohydrate diets. So, why would the body create something it has just spent a fair amount of effort to break down?
Certain key organs, including the brain, can use only glucose as an energy source. Therefore, it is essential that the body maintain a minimum blood glucose concentration. When the blood glucose concentration falls below that certain point, new glucose is synthesized by the liver to raise the blood concentration to normal.
Gluconeogenesis is not simply the reverse of glycolysis. There are some important differences. Pyruvate is a common starting material for gluconeogenesis. First, the pyruvate is converted into oxaloacetate. Oxaloacetate then serves as a substrate for the enzyme phosphoenolpyruvate carboxykinase (PEPCK), which transforms oxaloacetate into PEP. From this step, gluconeogenesis is nearly the reverse of glycolysis.
As will be discussed in a future lesson that addresses lipolysis, fats can be broken down into glycerol, which can be phosphorylated to form dihydroxyacetone phosphate, or DHAP. DHAP can either enter the glycolytic pathway or be used by the liver as a substrate for gluconeogenesis.
IN CONTEXT
Aging and the…
Body’s Metabolic Rate
The human body’s metabolic rate decreases nearly 2% per decade after age 30. Changes in body composition, including reduced lean muscle mass, are mostly responsible for this decrease. The most dramatic loss of muscle mass, and consequential decline in metabolic rate, occurs between 50 and 70 years of age. Loss of muscle mass is the equivalent of reduced strength, which tends to inhibit seniors from engaging in sufficient physical activity. This results in a positive-feedback system where the reduced physical activity leads to even more muscle loss, further reducing metabolism.
There are several things that can be done to help prevent general declines in metabolism and to fight back against the cyclic nature of these declines. These include eating breakfast, eating small meals frequently, consuming plenty of lean protein, drinking water to remain hydrated, exercising (including strength training), and getting enough sleep. These measures can help keep energy levels from dropping and curb the urge for increased calorie consumption from excessive snacking.
Although these strategies are not guaranteed to maintain metabolism, they do help prevent muscle loss and may increase energy levels. Some experts also suggest avoiding sugar, which can lead to excess fat storage. Spicy foods and green tea might also be beneficial. Because stress activates cortisol release, and cortisol slows metabolism, avoiding stress, or at least practicing relaxation techniques, can also help.
SOURCE: THIS TUTORIAL HAS BEEN ADAPTED FROM OPENSTAX “ANATOMY AND PHYSIOLOGY 2E”. ACCESS FOR FREE AT OPENSTAX.ORG/BOOKS/ANATOMY-AND-PHYSIOLOGY-2E/PAGES/1-INTRODUCTION. LICENSE: CREATIVE COMMONS ATTRIBUTION 4.0 INTERNATIONAL.